Samia Elzwi1*, Akram Alabdli2
1Assistant professor, Department of Pharmacology,University of Benghazi, Libya
2Lecturer, Department of Laboratory Management,University of Benghazi,Libya
*Correspondence: Samia Elzwi, Assistant professor,Department of Pharmacology, University of Benghazi,Libya, Email: Samia.Alzwi@uob.edu.ly
Received: 11 Dec, 2025; Accepted: 26 Dec, 2025; Published: 01 Jan, 2026.
Citation: Samia Elzwi and Akram Alabdali. “Effect of Syzygium Aromaticum (Clove) on Alloxan Induced Diabetes Mellitus in Mice.” J Nutr Diet Manage (2026):116. DOI: 10.59462/JNDM.4.1.116
Copyright: © 2026 Samia Elzwi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Diabetes mellitus is a common metabolic disease that is typically linked to insulin resistance and hyperglycemia. Diabetes mellitus is classified into type 1 diabetes mellitus due to idiopathic and autoimmune cause and type 2 that is due to insulin resistance. In addition, there is type 3 diabetes mellitus due to drugs like glucocorticoids and diazoxide and type 4 diabetes mellitus due to hormonal induced insulin resistance. Natural herbs have been used in treatment of diabetes mellitus due to less side effects and ease to getting the herbs One of the commonly used herbs is clove (Syzygium aromaticum) is used as flavoring agents in food in addition to its commonly used as dental analgesic. The paper showed that clove has glucose lowering effect in dose dependent manner mainly due to antioxidant properties of eugenol in addition to its flavonoid´s antidiabetic modulating action raises insulin secretion and GLUT4 translocation while decreasing apoptosis and insulin resistance.
Keywords
Alloxan, Clove and Diabetes mellitus, Herbs in Diabetes mellitus, Insulin.
Introduction
Diabetes mellitus is a common metabolic disease that is typically linked to insulin resistance and hyperglycemia.A disruption in insulin function or aberrant insulin secretion is the cause of hyperglycemia [1].Diabetes mellitus is classified into type 1 diabetes mellitus due to idiopathic and autoimmune cause and type 2 that is due to insulin resistance. In addition, there is type 3 diabetes mellitus due to drugs like glucocorticoids and diazoxide and type 4 diabetes mellitus due to hormonal induced insulin resistance. [2]. Diabetes mellitus in treated by different types of drugs like oral hypoglycemic drugs (sulfonylurea and meglitinide) and biguanides (metformin) and alpha glucosidase inhibitors (acarbose), peptide analog DPP4 inhibitors and sodium -glucose cotransport inhibitors and different types of insulin, each type of drugs acting by different mechanism and associated with many adverse effects [3]. Moreover, many types of natural remedy have been used in treatment of diabetes mellitus for many years like zin-giber officinale and cinnamon due to their antioxidant properties, in addition to clove that has hypoglycemic action due to its flavonoids [4]. Alloxan induced diabetes mellitus is common method for induction of diabetes in mice. Hyperglycemia and decreased insulin production result from alloxan’s targeting and destruction of pancreatic beta cells[5]. Type 1 diabetes and the mechanisms underlying complications from the disease are fequently studiedusing this model Natural herbs have been used in treatment of diabetes mellitus due to less side effects and ease to getting the herbs One of the commonly used herbs is clove (Syzygium aromaticum) is used as flavoring agents in food in addition to its commonly used as dental analgesic [6]. However, clove also commonly used during covid – 19 attacks. Many constituents in clove like eugenol and polyphenol have antioxidant properties therefore commonly used to lower blood glucose level. [7] Eugenol, its main constituent, is responsible for its antioxidant, antimicrobial, antinociceptive, antiviral, and anesthetic qualities, which give it a wide range of medical applications.
However, the flavonoids in clove reduce hyperglycemia in mice induced by both alloxan and streptozotocin diabetes mellitus. Flavonoids’ antidiabetic modulating action raises insulin secretion and GLUT4 translocation while decreasing apoptosis and insulin resistance [8]. The administration of clove essential oil (Syzygium aromaticum) will significantly reduce blood glucose levels in STZ-induced diabetic rats due to its antioxidant properties and potential insulin sensitizing effects, thereby demonstrating its efficacy as a therapeutic agent for the management of diabetes mellitus. [9] The aim of this paper is to study the effect of clove on alloxan induced diabetes mellitus in mice.

Figure 1: Chemical structure of some compound found in the clove

Figure 2: Syzygium aromaticum (Clove)
Material and Methods
Experimental Animals: Albino mice of either sex weighing 25-35g maintained in the animal house of Faculty of Medicine University of Benghazi, Libya. The mice were bred in the faculty animal house. All animals were housed in standard polypropylene cages (48×35×22cm) and kept under controlled room temperature (20±5 °C; relative humidity 60-70%) in a 12h light-dark cycle. The animals were given a standard laboratory diet and free water. Food was withdrawn 12h before and during the experimental hours The mice are divided into four groups each contain six mice
Methodology
Thirty male rats were divided into five groups (n = 6): Alloxan (150 mg/kg), Clove extract (250 and 500 mg/kg), Glibenclamide (5 mg/kg) and Control. Diabetes was induced using alloxan, and treatments were given orally daily. Blood glucose levels were measured and analyzed using descriptive statistics, Shapiro–Wilk test, Levene’s test, one-way ANOVA, and Games–Howell post hoc test, with significance set at p < 0.05.
Descriptive Analysis
Descriptive statistics were used to summarize blood glucose levels across experimental groups, expressed as mean ± SD. This provided an initial comparison between the alloxan-induced diabetic group and the treatment groups (clove extract, glibenclamide, and control), offering preliminary insights into the hypoglycemic effects of clove extract relative to the standard drug.
| Group | N | (Mean) | SD |
|---|---|---|---|
| Alloxan 150 mg/kg | 6 | 298.83 | 87.36 |
| Clove 250 mg/kg | 6 | 70.50 | 8.29 |
| Clove 500 mg/kg | 6 | 55.17 | 9.02 |
| Control | 6 | 65.33 | 6.44 |
| Glibemclamide 5mg/kg | 6 | 77.33 | 12.14 |
Table 1:Descriptive statistics (Mean ± SD) of blood glucose levels across experimental groups
According to the descriptive analysis we can see the group that received Alloxan had significantly higher blood glucose levels than any other group. Glibenclamide, clove extract (250 and 500 mg/kg) and the control group. On the other hand, showed significantly reduced glucose levels. The greatest glucose-lowering impact was shown by the higher dose of clove (500 mg/kg) which may indicate dose-dependent hypoglycemic action. As seen in table 1 and figure3.

Figure 3:Represent descriptive statistics (Mean ± SD) of blood glucose levels across experimental groups
Percentage reduction in blood glucose levels for each treatment group compared to the Alloxan group was calculated using the formula
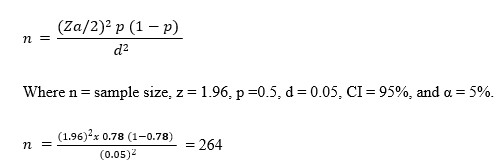
The hypoglycemic effectiveness of clove extract and the common medication (glibenclamide) can be compared to the hyperglycemic control using this formula which offers a straightforward and consistent way to measure the relative drop in glucose levels brought on by each therapy.
| Group | Mean ± SD | % Reduction (Alloxan) |
|---|---|---|
| Alloxan 150 mg/kg | 298.83 ± 87.36 | 298.83 ± 87.36 |
| Clove 250 mg/kg | 70.50 ± 8.29 | 70.50 ± 8.29 |
| Clove 500 mg/kg | 55.17 ± 9.02 | 55.17 ± 9.02 |
| Control | 65.33 ± 6.44 | 65.33 ± 6.44 |
| Glibenclamide 5 mg/kg | 77.33 ± 12.14 | 77.33 ± 12.14 |
Table 2:Represent that All treatment groups had significant drops in blood glucose levels as compared to the Alloxan group.
Table 2 Represent that All treatment groups had significant drops in blood glucose levels as compared to the Alloxan group.The greatest reduction (81.54%) was seen in the 500 mg/kg clove extract, which was closely followed by the 250 mg/kg clove group (76.41%) and the control group (78.14%). Clove extract, especially at the higher dose, has a substantial hypoglycemic impact comparable to the conventional medication, as seen by the slightly smaller reduction (74.12%) that glibenclamide generated compared to the clove-treated groups.
Hypothesis and One-Way ANOVA Analysis of Blood Glucose Levels
Our research examines how two different dosages of clove extract affect blood glucose levels in rats with Alloxan-induced hyperglycemia. One-way ANOVA can be used to test the hypothesis that the treatment groups’ mean blood glucose levels differ significantly from one another.
Statistical Testing
The previous part focused on the descriptive characteristics of the respondents. In this part of the study, we will go a step further to test the hypotheses of the study based on inference statistics.
Testing of Normality
There are two broad classifications of statistical tests used in data analysis, namely: parametric and non-parametric. The use of parametric tests is said to be appropriate when the variables distributed normally Conversely, non-parametric tests must appropriate
| Group | Mean ± SD % Reduction (Alloxan) | ||
|---|---|---|---|
| Statistic | df | Sig. | |
| Alloxan 150 mg/kg | 0.974 | 6 | 0.921 |
| Clove 250 mg/kg | 0.96 | 6 | 0.823 |
| Clove 500 mg/kg | 0.965 | 6 | 0.856 |
| Control | 0.897 | 6 | 0.359 |
| Glibenclamide 5 mg/kg | 0.84 | 6 | 0.131 |
Table 3:Shapiro-Wilk test of normality for blood glucose levels across experimental groups
The Shapiro–Wilk test results indicated that all groups exhibited non-significant p-values (p > 0.05), suggesting that the data are normally distributed within each treatment group. Therefore, the assumption of normality required for one-way ANOVA was satisfied, allowing for valid comparison of mean glucose levels across groups. see table 3.
| Levene Statistic | df1 | df2 | Sig. |
|---|---|---|---|
| 11.319 | 4 | 25 | .000 |
Table 4:Test of Homogeneity of Variances (Levene’s Test)
Table 4 show Levene’s test indicated a significant result (p < 0.05), suggesting that the assumption of homogeneity of variances was violated. Therefore, post hoc comparisons should be conducted using a method that does not assume equal variance
| Source | Sum of Squares | df | Mean Square | F | Sig. |
|---|---|---|---|---|---|
| Between Groups | 259369.53 | 4 | 64,842.383 | ||
| Within Groups | 39857.83 | 25 | 1,594.313 | 40.67 | .000 |
| Total | 299227.37 | 29 |
Table 5:One-Way ANOVA of Blood Glucose Levels
The one-way ANOVA revealed a highly significant difference in mean blood glucose levels among the experimental groups (F(4,25) = 40.67, p < 0.001), indicating that at least one group differed significantly from the other
| Comparison | Mean Difference (I–J) | Sig. | 95% CI (Lower–Upper) |
|---|---|---|---|
| Alloxan vs. Clove 250 mg/kg | 228.33 | 0.007 | 85.64 – 371.03 |
| Alloxan vs. Clove 500 mg/kg | 243.67 | 0.005 | 101.04 – 386.29 |
| Alloxan vs. Control | 233.50 | 0.006 | 90.66 – 376.34 |
| Alloxan vs. Glibenclamide | 221.50 | 0.008 | 79.21 – 363.79 |
| Clove 500 mg/kg vs. Glibenclamide | -22.17 | 0.035 | -42.82 – -1.51 |
Table 6:Post Hoc Multiple Comparisons of Blood Glucose Levels (Games–Howell Test)
Table 6 show the results of Games–Howell post hoc test revealed that the Alloxan group had significantly higher blood glucose levels compared to all other groups (p < 0.01). Additionally, clove extract at 500 mg/kg showed a significant reduction in glucose compared to glibenclamide (p=0.035). No other pairwise comparisons reached statistical significance, indicating that the hypoglycemic effect of clove extract at both doses is comparable to the control and close to the standard drug

Figure 4:Representation of the mean values for the different group
Discussion
Diabetes mellitus is common metabolic disease associated with insulin resistance and hyperglycemia The table (1) and figure (3) According to the descriptive analysis we can see the group that received Alloxan had significantly higher blood glucose levels than any other group. Glibenclamide, clove extract (250 and 500 mg/kg) and the control group. On the other hand, showed significantly reduced glucose levels. The greatest glucose-lowering impact was shown by the higher dose of clove (500 mg/kg) which may indicate dose-dependent hypoglycemic action. Clove reduced blood glucose in mice in dose dependent manner this is in accordance with previous study done by [10].Clove essential oil’s impact on blood sugar levels. treatment with clove essential oil resulted in a significant (P < 0.05) drop in glucose levels when compared to the positive control group. The groups that received 250 mg/kg of clove essential oil showed the biggest drop in blood sugar levels. A dose-dependent effect of clove essential oil on glucose levels was demonstrated by the smaller reduction observed in animals treated with 100 mg/kg of conventional diabetic medications as opposed to those treated with 100 mg/kg of clove essential oil. As shown in the table( 2) percentages of reduction in the blood glucose is greatest reduction (81.54%) was seen in the 500 mg/kg clove extract, which was closely followed by the 250 mg/kg clove group (76.41%) and the control group (78.14%). Clove extract, especially at the higher dose, has a substantial hypoglycemic impact comparable to the conventional medication, as seen by the slightly smaller reduction (74.12%) that glibenclamide generated compared to the clove-treated groups this is in line with study done in [11] showed that the therapeutic groups (clove oil, extract and powder) statistics had low levels of fasting blood sugar, total cholesterol, LDL-c, VLDL-c, triglycerides, AST, ALT, urea, uric acid, creatinine, bilirubin and MDA in comparison with the DC group. Contrarily, serum insulin levels, HDL-c, SOD and GSH actually were higher in the clove oil, extract and powder groups compared to the DC group. It could be concluded that the Clove oil, extract, or powder has demonstrated antioxidant properties and may be utilized as a functional food ingredient to guard against diabetes-related problems. Furthermore, the Shapiro–Wilk test results indicated that all groups exhibited non-significant p-values (p> 0.05), suggesting that the data are normally distributed within each treatment group. Therefore, the assumption of normality required for oneway ANOVA was satisfied, allowing for valid comparison of mean glucose levels across groups.as shown in the table (3) this is in line with study on The results with test rats indicated that alloxan administration induced the elevation of glucose level, activity of some liver enzymes, malondialdehyde (MDA), level of some lipid , urea and creatinine concentration are decreases
While the level of insulin, total thyroid hormones triiodothyronine (T3) and thyroxine (T4), glutathione content (GSH) in addition to the concentration of high density lipoprotein (HDL-C) were decreased under the effect of alloxan.
Rats with diabetes treated with either raw or irradiation clove extract showed improvements in hyperglycemia, hepatic, renal, and endocrine problems brought on by al loxn Clove extract exerts a dietary antidiabetic benefit and affect a partial normalization of the biochemical changes associated with alloxan-induced diabetes mellitus in rats. Furthermore, the study indicated that treatment of cloves with 10 kGy of gamma irradiation had no significant effect on the antidiabetic properties of the extract made from such cloves. This confirms the suitability of irradiation for the improvement of the hygienic quality of cloves. [12]. In addition clove has been reported to modulate physiological responses in diabetic rats, leading to a reduction in glucose levels, attributed to the antioxidant and hypoglycemic effects of clove essential oil components, particularly eugenol and eugenyl acetate [13]. These compounds facilitate glucose utilization through pathways independent of pancreatic function [14]. Hyperglycemia and hyperlipidemia are frequent side effects of diabetes, a chronic illness [15]. This study’s findings, which are consistent with those of Kuroda et al. showing the hypoglycemic effects of S. aromaticum [16], showed that clove improves glycemic control by raising serum insulin levels while lowering glucose levels. Another study’s findings suggested that S. aromaticum might inhibit the expression of the genes for “glucose 6-phosphatase” and “phosphoenolpyruvate car boxykinase,” both of which are known to have an enzymatic function in gluconeogenesis. As a result, S. aromaticum has effects similar to those of insulin and lowers the need for insulin by decreasing intestinal alpha-glucosidase enzyme activity and, consequently, glucose absorption [17]. According to clove’s hypoglycemic effects may be caused by a reduction in intestinal alpha-glucosidase activity, which is involved in intestinal glucose absorption [18]. The administration of clove essential oil (Syzygium aromaticum) will significantly reduce blood glucose levels in STZ-induced diabetic rats due to its antioxidant properties and potential insulin sensitizing effects, thereby demonstrating its efficacy as a therapeutic agent for the management of diabetes mellitus.The research conducted by [19] highlighted that diabetes mellitus is characterized by impaired carbohydrate, fat, and protein metabolism due to insufficient insulin secretion or decreased tissue sensitivity to insulin. According to. [20] and [21], clove has been reported to modulate physiological responses in diabetic rats, leading to a reduction in glucose levels, attributed to the antioxidant and hypoglycemic effects of clove essential oil components, particularly eugenol and eugenyl acetate [22]. These compounds facilitate insulin secretion.
References