Gang Badung Dalyop1*, Ogbonna Abigail Ify2 and Nnebechukwu Ijeoma Adaku2
1Department of Environmental Health, Federal College of Veterinary and Medical Laboratory Technology, NVRI, Vom.
2Department of Plant Science and Biotechnology, University of Jos.
*Correspondence: Gang Badung Dalyop, Department of Environmental Health, Federal College of Veterinary and Medical Laboratory Technology, NVRI, Vom, Email: gangbadung@gmail.com
Received: 19 Nov, 2025; Accepted: 26 Dec, 2025; Published: 31 Dec, 2025.
Citation: Gang BD, Ogbonna AI and Nnebechukwu IA. “Nutritional Enhancement of Fermented Digitaria Exilis L. (Acha) and Eleucine Coracana L. (L) Gaertn (Finger Millet) for Food Security and Health Benefits” J Nutr Diet Manage (2025):115. DOI: 10.59462/JNDM.3.1.115
Copyright: © 2025 Gang Badung Dalyop. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Fermented food products contribute to diet diversity globally which are vital to food security and significantly contribute to nutrition. The aim of this study was to evaluate the effects of fermentation on the nutritional and antinutritional composition of Digitaria exilis and Eleusine coracana flours. Acha and finger millet were processed into flours and subjected to fermentation in which 2000g of the samples was used for the evaluation. Proximate composition result revealed that Crude protein, ash and moisture contents increased while fat, carbohydrates and fibre decreased after fermentation for Acha and finger millet samples respectively. Carbohydrate showed highest mean values 77.80% for non-fermented and 76.02% for fermented samples of Acha, 74.70% and 73.83% for non-fermented and fermented finger millet. Results of vitamins indicated that vitamin A (19.30 mg/l) exhibited the highest mean values and amino acids portrayed that glutamic acid had 15.89g/100g as the highest mean value while tryptophan had 1.23g/100g as the lowest mean value. The mean values recorded for non-nutritional were significantly (p<0.05) different and flavonoids showed 2.75mg/100ml as the highest mean value for non-fermented while alkaloids have 0.59mg/100ml as the lowest mean value for fermented acha sample. The result of the minerals demonstrated that only Calcium, sulphur and Barium were increased after fermentation. Due to high nutritional contents of acha and finger millet flours, they can be use to make pudding, biscuits and other products.
Keywords
Fermented Foods, Nutrition, Antinutritional, Proximate, Health, Food Security
Introduction
Cereals serve as vital sources of macronutrients and en ergy for humans. Acha and Finger millet are traditional African cereals belonging to the Gramineae family, culti vated in West Africa for over 5000 years. They are com-monly grown in regions with low rainfall, particularly in the Plateau and savannah areas [1]. Acha is also known by various names such as hungry rice, petit mil, fundi grain, fonio rice, and “grain of life” [2,3]. The term “grain of life” stems from its ability to mature and be harvested within 3-4 months after sowing, providing food during the growing season when many other crops are still immature [4]. The two common varieties found in Africa are D. Exilis white acha) and D. iburua (black acha), with D. exilis being more prevalent in West Africa, specifically Nigeria, Ghana, and Benin [1]. In Nigeria, D. exilis is widely cultivated in Plateau State, as well as parts of Bauchi, Kebbi, Taraba, Kaduna, and Niger States. Acha is highly regarded for its nutritional value due to its composition and high concentration of macronutrients. It is rich in carbohydrates, di etary fiber, B-vitamins, and essential amino acids such as phenylalanine, tyrosine, methionine, and cystine [5], in ad dition to trace amounts of minerals like zinc, magnesium, manganese, iron, and potassium [6].Finger millet (Eleusine coracana L.) is one of the main mil lets grown in India. It is a rich source of minerals such as calcium, phosphorus, and iron contents [7]. The finger millet contains all essential amino acids such as cysteine, lysine, and methionine. Therefore, it can serve as an im portant source of vegetable proteins in the diet of vege tarian people. It also contains about 72% carbohydrates, including dietary fibre components and non-starchy poly saccharides which help in preventing constipation and help in decreasing the blood glucose level. It is rich in B-group vitamins such as riboflavin, thiamine, niacin, and folic acid [8]. The bran layers of finger millet comprise of phenolic contents, vitamins, and minerals, which provide numerous nutritional and therapeutic benefits [9]. It has nutraceutical properties and is recognized for its antidiabetic, anti-tum origenic, antidiarrheal, atherosclerogenic, anti-inflamma tory, antimicrobial, and antioxidant characteristics [10]. Finger millet is considered as the poor man’s food and can be stored for long period without being infested by insects and pests [11]. It is considered a gluten-free grain, with a lower glycemic index, and is generally used as whole grain flour for traditional food formulations, and can be utilized after processing in form of noodles, biscuits, muffins, ver micelli, pasta, and bread [12].
Materials and Methods
Sample Collection
Samples of Acha and finger millet for this work was ob tained from Vom Market, located in the Vwang District, within the Jos South Local Government Area of Plateau State. These samples were authenticated at the National Cereals Research Institute Outstation in Riyom Local Gov ernment Area of Plateau State before being transported to the Department of Plant Science and Biotechnology at the University of Jos for the purpose of flour production.
Sample Preparation
A weight of 1000 g of dried acha and finger millet samples were reduced to powder and used for the analysis.
Acha Grain: Cleaning (sorting) – Steeping (72 hrs) – oven drying (60oC, 24 hrs) – grinding –sieving (630 µm mesh size) – Acha flour.
Finger millet: Cleaning (sorting) – Steeping (72hrs) – oven drying (60oC, 24 hrs) –grinding – sieving (630 µm mesh size) – Acha flour.

(a) Digitaria exilis L. (Gilles, 2013)
Plate 1: Experimental Samples

(b) Eleucine coracana (L) Gaertn (Washira, 2015)
Determination of Moisture content This was determined using the AOAC [13] gravimetric method. The sample was thoroughly mixed. The water content was determined by weighing out 2g of the Sam ple into glass petri dish which has been previously dried and weighed. The dish including the samples were placed inside it in hot air oven for 5 hours at 130 ± 3o C. Finally, it was dried to constant weight, cool for ten minutes in a desiccator each time before weighing.
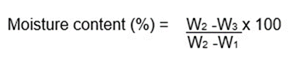
where,
W1 = weight of container with lid;
W2 = weight of container with lid and sample before dry ing; and W3 = weight of container with lid and sample after drying.
Determination of crude Protein
The nitrogen of protein and other compounds were con verted to ammonium sulphate by acid digestion with boil ing sulphuric acid using AOAC [14]. A known weight of the sample was placed in Kjeldahl flask and about 200 milligram of catalyst mixture (potassium sulphate, copper sulphate and selenium powder) was added. A measured volume of 10.0 cm3 of concentrated sulphuric acid was added to the content of the flask. It was gently heated for few minutes until frothing ceases and the heat was in creased to digest for 1 hour. It was allowed to cooled and made to a known volume with distilled water (100cm3). A measured volume of 10.0cm3 aliquot of the diluted solu tion of the digest was distilled by pipetting the volume into distillation chamber of micro Kjeldhal distillation apparatus and 10.0cm3 of 40% sodium hydroxide solution was add ed and steam distilled into 10.0cm3 of 4% boric acid con taining mixed indicator (colour from red-green was noted). The content was titrated with standard 0.01N hydrochloric acid to grey end point.
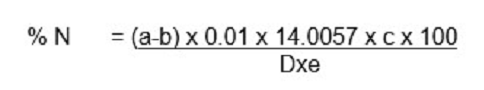 A = titre value for the sample
A = titre value for the sample
B = titre value for the blank
C = Volume to which digest is made up with distilled water
D = Aliquot taken for distillation
E = Weight of dried sample (mg)
To convert to % crude protein, it was multiplied by neces sary conversion factor (6.25).
Ash Determination
A measured weight of 2g test portion of the samples was weighed into porcelain crucible and place in muffle furnace preheated to 600oC using AOAC [14]. It was held at this temperature for 2 hours. The crucible was transferred di
rectly to desiccator, cooled and weighed immediately. It was reported in percentage to two decimal places.
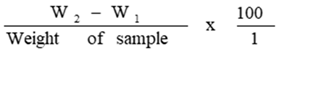
Ash (%) =
Where:
W1 = Weight (g) of empty crucible
W2 = Weight of crucible + Ash
Fat Determination (ether-extract)
A soxhlet extraction apparatus and 250ml quick fit flask which has been previously dried in the oven was fit up using AOAC [14]. A weight of 5g of the sample was trans ferred to a fat-free extraction thimble and slightly pluged with cotton wool. The thimble was placed in the extractor and 150cm3 of petroleum ether (B.P. 40-60oC) was added into the flask until it siphons over once. The source of heat (electrothermal heating mantle) was adjusted so that the ether boils gently and was left to siphon over for at least 6 hours. The flask (which now contains all the oil) was de
tached. The extract (oil) was filtered through Whatman fil ter paper into weighed beaker. The paper was finally wash with small portion of hot fresh ether. The solvent was evap orated at 100 degrees centigrade and beaker containing residue in an air oven was dried for 1 hour at 100-105 degree centigrade. The result was recorded as % oil to second decimal place.
Fat Determination = weight of oil× 100
Weight of biological material
Crude Fibre Determination
Defatted ground sample from fat determination was trans ferred into 250ml quick fit flask. 150ml of 1.25% sulphuric acid was added and fitted to reflux condenser. It was re fluxed for 30 minutes, cooled and filtered using Buchner
funnel fitted with Whatman filter paper. It was rinsed three times with hot distilled water, dried and the residue was carefully transferred into quick fit flask. A volume of 50ml of 1.25% Sodium hydroxide was added and refluxed for 30 minutes. It was filtered using Buchner funnel and rinsed three times with hot distilled water, once with 1.25% sulphuric acid and finally with 95% ethanol. The filter paper containing residue into porcelain crucible was removed and dried in an oven for 2 hours at 130oC. It was cooled in a desiccator, ash at 550 ± 10oC in muffle furnace, cooled in desiccator and weighed. This was determined by AOAC [14].
Crude fibre % = W2 -W3 x 100
Weight of sample
Where W2 = weight of crucible + Sample after washing, boiling and drying.
Carbohydrate
When the sum of the amounts of moisture, ash, crude pro tein, crude fat and crude fibre expressed in percentages was subtracted from 100, the difference was designated as the nitrogen-free extractives (NFE).
Carbohydrates = 100 - (% Moisture + % Proteins + % Lip ids + % Ash + % Fibres).
Vitamins
All vitamins were determined using AOAC 2019, Kirks and Sawyer’s methods.
Amino acids
Standard method of AOAC [15]. with some modification was used to determined amino acid profile in all the sam ples.
Mineral analysis
The standard wet digestion method as described by Tasie and Gebreyes [16] was utilized. In this approach, 0.5 g of acha flour was subjected to digestion using a mixture of 5ml concentrated nitric acid (HNO) and 1ml concentrated perchloric acid (HClO). After digestion, the sample was filtered and then brought up to a final volume of 100 ml in a standard flask. The XRF machine was employed to ana lyze all the minerals.
Anti-nutritional factors of experimental plant Anti-nutritional factors are substances that lower food in take or nutrient utilization/bioavailability. In actuality, they have a significant impact on restricting the broader application of numerous plants. These consist of cyanogenic, phytate, alkaloids, tannins, oxalate, and saponins. Using the double extraction gravimetric method outlined by Har borne [17], the sample’s saponin content was ascertained. The sample’s phytate content was ascertained using the procedure described by Lucas and Markakes [18]. The AOAC [13] methods were used to determine the sample’s tannin content. However, the approach was somewhat altered. The modified method of Abeza et al. [19] was used to determine the amount of oxalate in the powdered sam ple. The gravimetric method of Harborne [17] was used to determine the alkaloid content of the samples. Cyanide and steroids were determined using the method described by AOAC [14].
Statistical Analysis
All analytical determinations were conducted in triplicates. Data obtained were subjected to analysis of variance (ANOVA) using SPSS 23 version. Means and standard deviations were calculated. Duncan’s Multiple Range Test and T-Test was used for separation of means where significant differences existed.
Results and Discussion
Proximate Composition of Non-fermented and Fermented Acha and Tamba
In the current investigation, the results of the proximate analysis of both acha and finger millet in Table 1 showed an increased in the mean values of crude protein, ash con tent and moisture content while there was decreased in the mean values of fat, fibre and carbohydrates after fer mentation. The protein content of acha flour increased sig nificantly (p<0.05) from 10.91 to 11.95 upon fermentation in comparison to non-fermented flour. The maximum pro tein concentration was found in acha flour fermented by microorganisms, which was distinct from acha flours that was not fermented. This result supported the claims made by Olasupo and Falade [20], Adegunwa and Aluko [21], Ejike and Nwachukwu [22] that fermentation can increase the protein content of foods. Protein is a vital component that the body needs for several physiological processes, such as tissue growth, healing, and maintenance [23,24]. According to Yarwood and Miller [25] and Ogodo et al. [26], the ability of the fermenting organisms to manufac ture proteins is responsible for the rise in protein content during fermentation. Moreover, a larger protein content may also result from the fermentation-induced increase in microbial cell mass [27,28]. Other researches have re ported conclusions similar to this one. For example, during the fermentation of extruded mixes, Ojokoh and Onasanya [29] observed an increase in protein content. Likewise, Jeff-Agboola and Oguntuase [30] found that microorgan isms can raise the protein level in pearl millet. The high er protein content in fermented acha flour can contribute to its nutritional value and make it a more suitable ingre dient for functional foods and nutraceuticals. There was also a significant increase in crude protein contents of fin ger millet and the mean values increased significantly (P ≤ 0.05) from 10.11 to 10.89 during fermentation. The net production of protein components during the fermentation process of finger millet might have led to the synthesis of some amino acids resulting in increased protein contents during the fermentation process [31]. Inyang and Zakari [32] described that fermentation increased significantly (P ≤ 0.05) the protein contents of pearl millet flour. Hamad and Fidelis [33] reported that the protein content of finger millet increased significantly (P ≤ 0.05) from 11.56% to 12.31% after the fermentation treatment of grains. They stated that the increase in protein content could be credit ed to the utilisation of carbohydrate contents by the action of extracellular enzymes produced by the microorganisms involved in the fermentation as well as the degradation of complex proteins resulting in the release of the peptides and amino acids.
In this study, both non-fermented and fermented acha and finger millet flours showed an increase in ash con tent after 72 hours of fermentation. Mean values of ash of non-fermented acha increased from 1.00 to 1.58 after fermentation. Ndabikunze et al. [34] stated that the term “ash content” refers to the overall amount of minerals, in cluding potassium, calcium, and iron, that are present in food. Through enzymatic processes, lactic acid bacteria can liberate minerals from the substrate. These microor ganisms have the ability to decompose complex mineral forms and organic compounds, increasing their concentra tion in the fermented product and enhancing their bioavail ability [35,36]. In addition, fermentation can result in the degradation of phytic acid (through the activities of phy tase) which binds to minerals, preventing their absorption in the digestive system [26,37]. This can contribute to the higher ash content observed in the fermented acha flours. The increase in ash content during fermentation reported [26] in maize flour using a lactic acid bacteria consortium corroborates with the findings of the present study. There was a slight increase in ash content during fermentation treatments of finger millet and values increased from 2.09 to 2.88. No significant (P ≤ 0.05) difference in the ash content was seen in the fermented finger millet treatment.
Comparing the non-fermented flour to fermented flour, the results revealed a substantial (P<0.05) increase in mois ture content in the proximate composition of both acha flour and finger millet flour in the current study. According to this observation, the moisture content rises throughout fermentation processes from 6.91 to 7.08 for acha and from 7.50 to 7.75 for finger millet flour sample after fer mentation. [38-40], which suggest that a number of fac tors, including the addition of water to the substrate to promote microbial growth and activity, temperature, and drying time of the fermented acha flour, may be responsi-ble [41,42]. Regarding food quality, storage, and microbi ological safety, moisture content is a crucial factor to take into account. It is important to use appropriate drying and storage techniques.
There was a significant (P<0.05) decline in the fat content of the analysed samples and values diminished from 2.18 to 1.86 for acha and from 1.79 to 1.45 for finger millet after fermentation. The result is in conformity with the findings of Antony et al. [9] who reported about 42.9% reduction in the total fat content of the fermented sample. The observation in the current work agrees with the Results of Adegbehingbe [43] who reported a decreased in fat con tent in fermented maize samples. Declined in fat contents during fermentation process was reported by El-Beltagi et al. [44] that it might be due to the fact that physiological as well as biological changes during fermentation involves the use of energy resulting in the utilisation of lipids for the production of energy. It could also be due to the break down of fatty acids as well as glycerol components by the fermenting microorganisms which improve the taste, aro ma and texture of fermented flour products. The low fat content observe in the fermented samples could help in improving the shelf life of the fermented samples prevent ing chances of rancidity and declining the energy value of the fermented samples. Crude fibre, as an indigestible component of food, plays an important role in providing fibre to the diet and pro moting healthy digestion. It aids in regulating physiolog ical functions and contributes to the overall health of the intestines [45,46]. In this present study, the fibre content of the fermented acha and finger millet flours were found to be significantly lower compared to the non-fermented flours. Specifically, the fibre content decreased from 1.12 in non-fermented acha flour to 0.81 in fermented acha flour and from 3.81 to 3.20 finger millet flour samples. Microorganisms have the ability to utilize fibre as a car bon source for their growth and metabolic activities [47]. During fermentation, these bacteria produce enzymes that can break down the complex fibre structures into simpler components, which they can then utilize for energy and growth. As a result, the fibre content of the fermented acha flour decreases [35]. These decrease in fibre contents observed in the present study aligns with the findings of Ogodo et al. [48], who reported a similar decrease in fibre content during the fermentation of Sorghum bicolor flour with a lactic acid bacteria consortium. These results are also consistent with the findings of [49] who reported that the fibre content of pearl millet flour got reduced from 2.0% to 1.8% in fermented flour of pearl millet.
Carbohydrates are vital nutrients that play a crucial role in various physiological functions, including supporting the immune system and facilitating human development [50]. They are one of the main types of nutrients and serve as the primary source of energy for the body. Additional ly, carbohydrates are a significant substrate for microbial fermentation [51]. In this research, a significant decrease (p<0.05) in the carbohydrate content was observed in fermented acha and finger millet flours compared to the non- fermented flour. According to [52], the reduction in carbohydrate content can be attributed to the utilization of fermentable sugars by lactic acid bacteria during the fer mentation process. Lactic acid bacteria metabolize these sugars for their own growth and other metabolic activities, which leads to a decrease in the overall carbohydrate con tent of the fermented product [52]. Similar findings were reported by [53] during the fermentation of breadfruit and cowpea blend flours, where a decrease in carbohydrate content was observed. They also attributed the decrease to the metabolic activities of microorganisms during fer mentation, as they consume and convert the carbohy drates present as substrate. [52,54] agreed that utilization of fermentable sugars by lactic acid bacteria for growth and metabolic activities is a common phenomenon in fer mentation processes.
| Proximate Composition | Samples | |
| Digitaria exilis (Acha) | Eleusine coracana (Tamba) | |
| Non-fermented | ||
| Crude Protein | 10.91b | 10.11b |
| Fat | 2.18d | 1.79e |
| Ash | 1.00e | 2.09e |
| Crude Fibre | 1.12e | 3.81d |
| Moisture content | 6.91c | 7.50c |
| Carbohydrate | 77.88a | 74.70a |
| SE+ | 0.117 | 0.131 |
| p-value | 0 | 0 |
| Fermented | ||
| Crude Protein | 11.95b | 10.89b |
| Fat | 1.86d | 1.45f |
| Ash | 1.58de | 2.88e |
| Crude Fibre | 0.81e | 3.20d |
| Moisture content | 7.08c | 7.75c |
| Carbohydrate | 76.02a | 73.83a |
| SE+ | 0.252 | 0.043 |
| p-value | 0 | 0 |
| Independent T-test (non-fermented x Fermented) | 0.990ns | 1.000ns |
| Values (in the same column) with the same subscript letters do not differ significantly from each other according to the Duncan multiple range test; WHERE: SE = Standard Error; ** = significant; ns = not significant | ||
Table 1: Proximate Composition (%) of Non-fermented and Fer mented Acha and Tamba
Vitamins in Non-fermented and Fermented Acha and Tamba
Fermentation has been reported to exhibit varying effects on different vitamins such as B vitamins and vitamin E in cereals and legumes. In most of the studies, especially in the fermentation of maize, buckwheat, rice and sorghum using different starter cultures (LAB species, yeast and fungi), an increase in vitamin B1, B2, B3 and E were reported by up to 10 folds [55-58]. [59] reported that vitamin B increased in fermented maize flour due to enzyme in teractions with starch, protein and other key biosynthet ic precursors, which stimulated their synthesis of bound forms of the vitamins. Contrary to these studies, [60,61], on the fermentation of maize and tea, reported a reduction in vitamin B1, B2, β-carotene (as retinol equivalent) and the folate content of the resulting flour and their products. A decrease in vitamins was caused as a result of mechan ical loss due to processes, fermentation and lipid solubili zation, as well as consumption by other microorganisms or losses due to discarding the supernatant. In the recent study, there was decreased in the mean values of all the Vitamins that were assessed for both non-fermented and fermented samples of acha and finger millet. Similar case was also observed in types of Vitamin B for both non-fer mented and fermented samples of acha and finger millet. For acha sample, Vitamin A, B C, D, E and K decreased from non-fermented to fermented as follows: 19.30-3.17, 13.82 – 7.01, 1.93 -1.01, 0.31 – 0.14, 2.30 – 0.07 and 1.70 -0.003. For finger millet sample, the decreased in the mean values were as follows: Vitamin A (8.01-1.97), B (12.98-9.24), D (1.90-0.71), E (1.80 -0.00), K (2,34-0.00) and there was no any change in vitamin C (1.93-1.93). In some other studies, fermentation reportedly increased the vitamin B1, B2 and E (α-tocopherols) levels of fermented legumes (cowpea and kidney beans) by 17 to 94% [62,63]. Likewise, levels of vitamin A, B1, B2, B3, α-, γ- and δ- to copherols reportedly reduced in fermented common bean, cowpea, lupin and mung bean by 5–106% [64,62,65,66]. The level of vitamins after fermentation seemed to be de pendent on the fermenting strain and metabolic activity of these strains. This could have impacted the varying report ed trends in the vitamin content.
| Vitamins | Samples | |
| Digitaria exilis (Acha) | Eleusine coracana (Tamba) | |
| Non-fermented | ||
| Vitamin A | 19.30a | 8.01b |
| Vitamin B | 13.82b | 12.98a |
| Vitamin C | 1.93d | 1.93d |
| Vitamin D | 0.31f | 1.90d |
| Vitamin E | 2.30c | 1.80e |
| Vitamin K | 1.70e | 2.34c |
| SE+ | 0.014 | 0.019 |
| p-value | 0 | 0 |
| Fermented | ||
| Vitamin A | 3.17b | 1.97b |
| Vitamin B | 7.01a | 9.24a |
| Vitamin C | 1.01c | 1.93b |
| Vitamin D | 0.14d | 0.17c |
| Vitamin E | 0.007e | 0.00c |
| Vitamin K | 0.003e | 0.00c |
| SE+ | 0.019 | 0.105 |
| p-value | 0 | 0 |
| Independent T-test (non-fermented x Fermented) | 0.017** | 0.053** |
| Values (in the same column) with the same subscript letters do not differ significantly from each other according to the Duncan multiple range test; WHERE: SE = Standard Error; ** = significant; ns = not significant | ||
Table 2:Vitamins Composition of Digitaria exilis and Eleusine coracana
Results on Table 3 show means of amino acid composition of the samples. The results reveal that there is significant difference (p<0.05) amongst the samples of non-fermented and fermented for both Digitaria exilis (Acha) and Eleusine coracana (Tamba). The T-test result on Digitaria exilis (Acha) for both samples (non-fermented and fermented) were not significantly (p>0.05) different (0.879; 0.684) respectively. [67] reported increase in total free amino acid during fermentation of acha and maize and attributed it to the proteolytic activities of fermenting organisms. In the case of fermented Acha, there was a minor rise in leucine (9.02 compared to 8.93), lysine (2.62 compared to 2.41), isoleucine (4.03 compared to 3.99), and glutamic acid (15.89 compared to 15.22). Nonetheless, these changes were not statistically significant, which aligns with the observations made by [68] and [69], who noted slight to moderate alterations in amino acids in fermented millet products depending on the strain, fermentation time, and pH levels. Acha showed minimal changes in amino acids that were not significant, while Tamba demonstrated more substantial increases in the same amino acids. This indicates that factors such as the type of cereal, the configuration of proteins, and the activity of microorganisms during fermentation influence the release and modification of certain amino acids. These findings support earlier research demonstrating that fermentation can enhance the nutritional quality of grains, particularly in terms of the bioavailability of essential amino acids [70,71].
| Amino Acids | Samples | |
| Digitaria exilis (Acha) | Eleusine coracana (Tamba) | |
| Non-fermented | ||
| Leucine | 8.93b | 9.34c |
| Lysine | 2.41j | 4.64i |
| Isoleucine | 3.99e | 4.22k |
| Phenylalanine | 4.97cd | 5.06h |
| Tryptophan | 1.23m | 1.39p |
| Valine | 4.91d | 5.73f |
| Methionine | 3.95e | 2.38o |
| Proline | 3.55g | 9.55b |
| Arginine | 3.87f | 5.51g |
| Tyrosine | 3.27i | 3.79m |
| Histidine | 2.01k | 2.81n |
| Cysteine | 1.82l | 0.36q |
| Alanine | 5.01c | 7.97d |
| Glutamic acid | 15.22a | 20.60a |
| Glycine | 3.42h | 4.35jk |
| Threonine | 3.44h | 4.03l |
| Serine | 3.24i | 4.51ij |
| Aspartic acid | 5.02c | 6.98e |
| SE+ | 0.025 | 0.059 |
| p-value | 0 | 0 |
| Fermented | ||
| Leucine | 9.02b | 9.66c |
| Lysine | 2.62i | 4.84h |
| Isoleucine | 4.03e | 4.62i |
| Phenylalanine | 5.05d | 5.50g |
| Tryptophan | 1.31k | 1.44n |
| Valine | 5.00d | 5.99f |
| Methionine | 4.01e | 2.40m |
| Proline | 3.65f | 10.25b |
| Arginine | 4.04e | 5.94f |
| Tyrosine | 3.44gh | 3.96k |
| Histidine | 2.11j | 3.04l |
| Cysteine | 2.06j | 0.48o |
| Alanine | 5.12d | 8.00d |
| Glutamic acid | 15.89a | 20.74a |
| Glycine | 3.51fg | 4.68hi |
| Threonine | 3.50g | 4.25j |
| Serine | 3.32h | 4.70hi |
| Aspartic acid | 5.36c | 7.01e |
| SE+ | 0.049 | 0.069 |
| p-value | 0 | 0 |
| Independent T-test (non-fermented x Fermented) | 0.879ns | 0.684ns |
| Values (in the same column) with the same subscript letters do not differ significantly from each other according to the Duncan multiple range test; WHERE: SE = Standard Error; ** = significant; ns = not significant | ||
Table 3: Amino Acids Composition in Non-fermented and Fermented Acha and Tamba flours
Elemental Composition of Non-Fermented and Fermented Acha and Tamba flours
The elemental composition of Non-fermented and fer mented Acha and Tamba is presented in Table 4 and 5 respectively. Fermentation was found to improve the min eral content of Ca, S and Ba in acha flour compared to non-fermented acha flour. This increase in mineral con tent is attributed to the breakdown of antinutritional fac tors, such as phatic acid and oxalate, during fermentation. Phytic acid and oxalate can bind with minerals and reduce their absorption and bioavailability in the body. By reduc ing these antinutrients, fermentation enhances the avail ability of minerals in the fermented acha flour. The study also revealed that fermentation decreased the quantity of most macro and trace minerals Mo, Zr, Sr, Rb, Zn, Fe and Nb. For the finger millet sample, the study revealed that fermentation decreased the quantity of all macro and trace minerals except for Barium (Ba). The decreased in the mineral elements could be due to the utilisation of the elements by the fermented microorganisms during fer mentation. The increase in other macro and trace minerals indicates that fermented acha is more desirable for meet ing dietary mineral needs. Similar findings were [38] in a study on fermented hungry rice, where they observed an increase in iron, sodium, zinc, magnesium, manganese, and potassium during the fermentation process. Addition ally, [72] reported an increase in manganese, zinc, and iron during the fermentation of soy milk by lactic acid bac teria. The consistent results from these studies emphasize the effectiveness of fermentation in improving the mineral content of various food products. This has significant im plications for improving the nutritional quality and overall health benefits of traditional staple foods like acha, making them more valuable dietary sources of essential minerals.
| Mineral Elements (ppm) | Samples | |
| Non-fermented | fermented | |
| Mo | 12.29± 0.13a | 6.47± 0.05b |
| Zr | 6.32± 0.04a | 5.67± 0.06b |
| Sr | 2.94± 0.05a | 1.51± 0.07b |
| Rb | 7.15± 0.10a | 0.00± 0.00b |
| Zn | 25.86± 0.09a | 0.00± 0.00b |
| Fe | 142.77± 0.06a | 77.08± 0.03b |
| Ca | 98.46± 0.01b | 205.10± 0.04a |
| S | 1522.28± 0.16a | 3824.25± 0.09a |
| Ba | 998103.91± 0.11b | 999880.27± 0.12a |
| Nb | 12.43± 0.01a | 8.09± 0.01b |
| L.S.D | 0.12 | |
| P-value | ≤0.0001 | |
Table 4: Elemental composition of Non-fermented and fer mented Acha flour
| Minerals (ppm) | Samples | |
| Non-fermented Tamba | Fermented Tamba | |
| Mo | 8.17±0.06a | 6.49±0.08b |
| Zr | 6.44±0.12a | 3.90±0.06b |
| Sr | 17.39 ±0.17a | 16.28±0.05b |
| Rb | 23.21 ±0.05a | 12.79±0.08b |
| Zn | 28.43 ±0.07a | 23.44±0.10b |
| Fe | 160.44 ±0.08a | 137.08±0.02b |
| Mn | 448.31 ±0.19a | 260.29±0.08b |
| SC | 35.37 ±0.10a | 28.48±0.11b |
| Ca | 8287.00 ±0.26a | 7980.92±0.33b |
| K | 13706.09 ±0.05a | 6734.36±0.13b |
| S | 1778.27 ±0.61ba | 715.10±0.72b |
| Ba | 982731.39 ±0.16a | 990693.32±0.15b |
| Nb | 8.37 ±0.04a | 6.84±0.04b |
| L.S.D | 36 | |
| P-Value | ≤ 0.0000 | |
| At P≤0.05 there was a significant difference in the Elemental composition of Non-fermented and Fermented Tamba. Values are presented as mean±standard error of means. Ranking was done across the Non-fermented and Fermented Tamba products and values with the same super script are not significant. | ||
Table 5: The Elemental composition of Non-fermented and fer mented Tamba
Non-Nutritional Components of Non-fer mented and fermented Acha and Tamba flours
Food fermentation has been shown to effectively increase the nutritional composition of foods as well as decrease the levels of antinutritional factors (ANFs) and toxic con stituents, and might be a better alternative in minimizing the adverse effects of these compounds in diets [73] and [74]. The effect of fermentation on the anti-nutrients such as oxalate, saponins, Alkaloids, flavonoids, cyanide, ste roids, aflatoxins, phytic acid, and tannin contents were also studied for both non-fermented and fermented Acha and Finger millet samples. The mean value recorded for each sample for non-nutritional were significantly (p<0.05) different for non-fermented and fermented of Digitaria exi lis (Acha) and Eleusine coracana (Tamba). The results are in line with the studies of Makokha et al. [75] who reported a reduction in the phytic acid content of finger millet after fermentation treatments.
| Non-Nutritional | Samples | |
| Digitaria exilis (Acha) | Eleusine coracana (Tamba) | |
| Non-fermented | ||
| Oxalate | 0.78d | 1.66b |
| Tannins | 0.21g | 1.65b |
| Phytate | 2.07b | 0.87f |
| Saponins | 0.56f | 0.55g |
| Alkaloids | 1.13c | 2.10a |
| Flavonoids | 2.75a | 0.96e |
| Cyanide | 0.003h | 1.00d |
| Steriods | 0.67e | 1.33c |
| SE+ | 0.012 | 0.012 |
| p-value | 0 | 0 |
| Fermented | ||
| Oxalate | 0.71c | 1.51d |
| Tannins | 0.14f | 1.55d |
| Phytate | 1.07a | 1.85c |
| Saponins | 0.003g | 0.32g |
| Alkaloids | 0.0003g | 1.99b |
| Flavonoids | 0.59d | 2.40a |
| Cyanide | 0.24e | 1.32e |
| Steriods | 0.99b | 0.15h |
| SE+ | 0.015 | 0.027 |
| p-value | 0 | 0 |
| Independent T-test (non-fermented x Fermented) | 0.028** | 0.199ns |
| Values (in the same column) with the same subscript letters do not differ significantly from each other according to the Duncan multiple range test; WHERE: SE = Standard Error; ** = significant; ns = not significant | ||
Table 6: Non-nutritional Components of Non-Fermented and Fermented Acha and Tamba
CONCLUSION
Fermentation has a significant impact on the proximate composition of acha and finger millet flours. It resulted in a significant increase in Crude protein, ash content and moisture while Crude fibre, fat content and carbohydrate Content decreased. Fermentation also led to reduction in non-nutritional factors. These changes contributed to the improved nutritional quality, safety, and potential health benefits of fermented Acha and finger millet flours. This study will help in the promotion of traditional processing techniques in enhancing the utilization of underutilized acha and finger millet by incorporating them for the devel opment of functional food products with lower antinutritional components, high nutritional value and better bioavail ability of micronutrients.
References
1. Wakil, S. M., and E. M. Olorode. “Potential probiot ic properties of lactic acid bacteria isolated from malt ed and spontaneously fermented acha (Digitaria exi lis) flour.” Advances in Research 17, no. 6 (2018): 1-12.
2. Jideani, I. A., and Victoria A. Jideani. “Developments on the ce real grains Digitaria exilis (acha) and Digitaria iburua (iburu).” Journal of food science and technology 48, no. 3 (2011): 251-259.
3. Karasu, A., H. C. Alpsoy, and M. Öz. “Effects of some natural origined growth regulating substanc es on the seed yield and some agronomical charac teristics of common pea (Pisum sativum).” Journal of Food and Nutrition Research 47, no. 3 (2015): 60-67.
4. Temple, Victor J., and James D. Bassa. “Proximate chemical composition of Acha (Digitaria exilis) grain.” Journal of the Science of Food and Agriculture 56, no. 4 (1991): 561-563.
5. Ayo, J. A., V. A. Ayo, I. Nkama, and R. Adewori. “Physiochemi cal, in-vitro digestibility and organoleptic evaluation of” Acha” wheat biscuit supplemented with soybean flour.” (2007).
6. Chukwu, Ogbonnaya, and Aminat Joy Abdul-Kadir. “Proximate chemical composition of acha (Dig itaria exilis and Digitaria iburua) grains.” Jour nal of Food Technology 6, no. 5 (2008): 214-216.
7. Narasinga Rao, B. S., Y. G. Deosthale, and K. C. Pant. “Nu tritive value of Indian foods.” Hyderabad, India: Indian Coun cil of Medical Research, National Institute of Nutrition (1989).
8. Vidyavati, H. G., J. M. Begum, J. Vijayakumari, S. S. Goka vi, and Shamshad Begum Shamshad Begum. “Utilization of finger millet in the preparation of papad.” (2004): 379-382.
9. Antony, Usha, G. Sripriya, and T. S. Chandra. “Ef fect of fermentation on the primary nutrients in fin ger millet (Eleusine coracana).” Journal of Agricultur al and Food Chemistry 44, no. 9 (1996): 2616-2618.
10. Chandra, Dinesh, Satish Chandra, and A. K. Sharma. “Review of Finger millet (Eleusine coracana (L.) Gaertn): A power house of health benefiting nutrients.” Food Sci ence and Human Wellness 5, no. 3 (2016): 149-155.
11. Erasmus, AISONI Japhet, M. Yushau, and OROLE Olu kayode Olugbenga. “Processing effects on physico chemical and proximate composition of finger millet (Eleusine coracana).” Greener J Biol Sci 8 (2018): 14.
12. Rathore, Teena, Rakhi Singh, Dinkar B. Kamble, Ashutosh Upadhyay, and S. Thangalakshmi. “Review on finger millet: Processing and value addition.” The Pharma Innovation Journal 8, no. 4 (2019): 283-291.
13. Cunniff, P., and D. Washington. “Official methods of analysis of AOAC International.” J. AOAC Int 80, no. 6 (1997): 127A.
14. Horwitz, W., and G. W. Latimer. “Official methods of analysis of AOAC International. 18th edGaithers burg.” AOAC International: Rockville, MD, USA (2005).
15. Papaneophytou, Christos. “Modern Applications in the Deter mination of Food and Feed Additives.” In Sustainable Use of Feed Additives in Livestock: Novel Ways for Animal Production, pp. 875-912. Cham: Springer International Publishing, 2023.
16. Tasie, Masresha Minuye, and Belay Gezahegn Gebreyes. “Characterization of nutritional, antinutritional, and mineral contents of thirty‐five sorghum varieties grown in Ethiopia.” In ternational journal of food science 2020, no. 1 (2020): 8243617.
17. Harborne, J. B. “Extraction and isolation of plant constituents. In Phytochemical methods: A guide to modern techniques of plant analysis”. Spring er Science & Business Media (1984) pp. 12-25.
18. Lolas, George M., and Pericles Markakis. “Phyt ic acid and other phosphorus compounds of beans (Phaseolus vulgaris L.).” Journal of Agricultur al and Food Chemistry 23, no. 1 (1975): 13-15.
19. Abaza, R. H., J. T. Blake, and E. J. Fisher. “Oxalate de termination: Analytical problems encountered with cer tain plant species.” Journal of the association of of ficial analytical chemists 51, no. 5 (1968): 963-967.
20. Yang, Hui, Yezhi Qu, Jiantao Li, Xianqi Liu, Rina Wu, and Junrui Wu. “Improvement of the protein quality and degra dation of allergens in soybean meal by combination fermen tation and enzymatic hydrolysis.” Lwt 128 (2020): 109442.
21. Adegunwa, M. O., and R. E. Aluko. “Fermentation and enzymatic treatments for improving protein content and quality of plant-based foods.” Critical Reviews in Food Science and Nutrition 59, no. 16 (2019): 2549-2562.
22. Iheanacho, Chinedu Christian. “AND MINERAL COM POSITIONS of Digitaria exilis Alloysius Chibuike Ogodo’’*, Joy Otabor”, Chrinius Hammuel’.” (2024).
23. Pasiakos, Stefan M., Tom M. McLellan, and Harris R. Lieb erman. “The effects of protein supplements on muscle mass, strength, and aerobic and anaerobic power in healthy adults: a systematic review.” Sports medicine 45, no. 1 (2015): 111-131.
24. Wolfe, Robert R. “Branched-chain amino acids and muscle protein synthesis in humans: myth or reality?.” Journal of the International Society of Sports Nutrition 14, no. 1 (2017): 30.
25. Ogodo, Alloysius Chibuike, Joy Otabor, Chrinius Hammuel, and Chinedu Christian Iheanacho. “STUDIES ON THE EF FECTS OF LACTIC ACID BACTERIA FERMENTATION ON THE PROXIMATE AND MINERAL COMPOSITIONS of Digi taria exilis.” Journal of Experimental Research 12, no. 1 (2024).
26. Ogodo, Alloysius Chibuike, Ositadinma Chinyere Ugbogu, Reginald Azu Onyeagba, and Hope Chukwuemeka Oker eke. “Effect of lactic acid bacteria consortium fermentation on the proximate composition and in-vitro starch/protein digestibility of maize (Zea mays) flour.” American journal of microbiology and biotechnology 4, no. 4 (2017): 35-43.
27. Ogodo, Alloysius Chibuike, Ositadinma Chinyere Ug bogu, Reginald Azu Onyeagba, and Hope Chukwue meka Okereke. “In-vitro starch and protein digest ibility and proximate composition of soybean flour
fermented with lactic acid bacteria (LAB) consortia.” Agri culture and Natural Resources 52, no. 5 (2018): 503-509.
28. Rezac, Shannon, Car Reen Kok, Melanie Heermann, and Robert Hutkins. “Fermented foods as a dietary source of live organisms.” Frontiers in microbiology 9 (2018): 1785.
29. Ojokoh, A. O., and T. T. Onasanya. “Effect of fermentation and extrusion on the nutrient and anti-nutrient composition of soy beans (Glycine max, L) and Acha (Digitaria exilis Stapf).” Micro biology Research Journal International 21, no. 1 (2017): 1-21.
30. Jeff-Agboola, Y. A., and O. S. Oguntuase. “Effect of Bacillus sphaericus on proximate composition of soy bean (Glycine max) for the production of soy iru.” Pa kistan Journal of Nutrition 5, no. 6 (2006): 606-607.
31. Uwaegbute, A. C., C. U. Iroegbu, and O. Eke. “Chemical and sensory evaluation of germinated cowpeas (Vigna unguiculata) and their products.” Food chemistry 68, no. 2 (2000): 141-146.
32. Inyang, C. U., and U. M. Zakari. “Effect of germination and fermentation of pearl millet on proximate, chemical and sensory properties of instant” Fura”-a Nigerian cereal food.” Pakistan journal of Nutrition 7, no. 1 (2008): 9-12.
33. HAMAD, AHMED M., and Ml L. FIELDS. “Evaluation of the pro tein quality and available lysine of germinated and fermented cereals.” Journal of food science 44, no. 2 (1979): 456-459.
34. Boudaoud, Sonia, Delphine Sicard, Lucas Suc, Gen eviève Conéjéro, Diego Segond, and Chahinez Aouf. “Ferulic acid content variation from wheat to bread.” Food Science & Nutrition 9, no. 5 (2021): 2446-2457.
35. Tamang, Jyoti P., Dong-Hwa Shin, Su-Jin Jung, and Soo Wan Chae. “Functional properties of microorganisms in fermented foods.” Frontiers in microbiology 7 (2016): 578.
36. Dahiya, Dinesh K., Renuka, Monica Puniya, Umesh K. Shandilya, Tejpal Dhewa, Nikhil Kumar, Sanjeev Kumar, Anil K. Puniya, and Pratyoosh Shukla. “Gut microbiota modula tion and its relationship with obesity using prebiotic fibers and probiotics: a review.” Frontiers in microbiology 8 (2017): 563.
37. Yi, Ruokun, Yu Tian, Fang Tan, Wenfeng Li, Jian fei Mu, Xingyao Long, Yanni Pan, and Xin Zhao. “In tervention effect of Malus pumila leaf flavonoids on senna‐induced acute diarrhea in BALB/c mice.” Food Science & Nutrition 8, no. 5 (2020): 2535-2542.
38. Echendu, C. A., I. C. Obizoba, J. U. Anyika, and P. C. Ojimelukwe. “Changes in chemical composition of treat ed and untreated hungry rice “Acha”(Digitaria exilis).” Pa kistan journal of nutrition 8, no. 11 (2009): 1779-1785.
39. Uwagbale, Edward-Ekpu Douglas, Audu Stephen Saratu, Odiba Victor Akagwu, Okopi Ochaba Ste phen, and Affiku Manyi Lilian. “African cereals and non-African cereals: a comparative review of their nu tritional composition.” World 1, no. 2 (2016): 30-37.
40. Mustapha, K. B., H. L. Zubairu, and A. Adamu. “Compar ison of nutritional values of wheat (Triticum aestivum) and acha (Digitaria exilis) grains.” Bayero Journal of Pure and Applied Sciences 11, no. 1 (2018): 133-138.
41. Abdalla, A. A., A. H. El Tinay, B. E. Mohamed, and A. H. Abdalla. “Proximate composition, starch, phy tate and mineral contents of 10 pearl millet geno types.” Food chemistry 63, no. 2 (1998): 243-246.
42. de Souza, Vanessa Rosse, Adriana Aniceto, Joel Pimentel Abreu, Julia Montenegro, Bruno Boquimpani, Vanessa Aze vedo de Jesuz, Monique de Barros Elias Campos et al. “Fruit‐ based drink sensory, physicochemical, and antioxidant prop erties in the Amazon region: Murici (Byrsonima crassifolia (L.) Kunth and verbascifolia (L.) DC) and tapereba (Spondia mombin).” Food science & nutrition 8, no. 5 (2020): 2341-2347.
43. Adegbehingbe, Kehinde Tope. “Fermented sprouted and unsprouted maize for ogi production.” International Jour nal of advanced research 1, no. 10 (2013): 428-434.
44. El-Beltagi, Hossam Saad, Amal Amin Mohamed, and Ba haa El-Din Bastawy Mekki. “Differences in some constitu ents, enzymes activity and electrophoretic characterization of different rapeseed (Brassica napus L.) cultivars.” An nals of Oradea University, Biology Fascicle/Analele Uni versităţii din Oradea, Fascicula Biologie 18, no. 1 (2011).
45. Iheanacho, Chinedu Christian. “AND MINERAL COM POSITIONS of Digitaria exilis Alloysius Chibuike Ogodo’’*, Joy Otabor”, Chrinius Hammuel’.” (2024).
46. Iheanacho, Chinedu Christian. “AND MINERAL COM POSITIONS of Digitaria exilis Alloysius Chibuike Ogodo’’*, Joy Otabor”, Chrinius Hammuel’.” (2024).
47. Iheanacho, Chinedu Christian. “AND MINERAL COM POSITIONS of Digitaria exilis Alloysius Chibuike Ogodo’’*, Joy Otabor”, Chrinius Hammuel’.” (2024).
48. Ogodo, Alloysius Chibuike, Dawn Ify Agwaranze, Nkechi Val entina Aliba, Adindu Chukwuma Kalu, and Chioma Blessing Nwaneri. “Fermentation by lactic acid bacteria consortium and its effect on anti-nutritional factors in maize flour.” (2019): 17-23.
49. Sade, Fasasi Olufunmilayo. “Proximate, antinutritional factors and functional properties of processed pearl millet (Pennisetum glaucum).” Journal of food technology 7, no. 3 (2009): 92-97.
50. Chatterjee, R., and H. C Yeh. “Edible carbohydrates: A review of their digestion, absorption, and phys iological utilization”. Critical Reviews in Food Sci ence and Nutrition 58 no 14 (2018): 2371-2378.
51. Ojokoh, A. O., O. E. Fayemi, F. C. K. Ocloo, and F. I. Nwoko lo. “Effect of fermentation on proximate composition, phys icochemical and microbial characteristics of pearl millet (Pennisetum glaucum (L.) R. Br.) and Acha (Digitaria exilis (Kippist) Stapf) flour blends.” Journal of Agricultural Biotech nology and Sustainable Development 7, no. 1 (2015): 1-8.
52. Iheanacho, Chinedu Christian. “AND MINERAL COM POSITIONS of Digitaria exilis Alloysius Chibuike Ogodo’’*, Joy Otabor”, Chrinius Hammuel’.” (2024).
53. Ojokoh, A. O., M. K. Daramola, and O. J. Oluoti. “Ef fect of fermentation on nutrient and anti-nutrient com position of breadfruit (Treculia africana) and cowpea (Vigna unguiculata) blend flours.” African Journal of Agricultural Research 8, no. 27 (2013): 3566-3570.
54. Ogodo, Alloysius Chibuike, Joy Otabor, Chrinius Hammuel, and Chinedu Christian Iheanacho. “STUDIES ON THE EF FECTS OF LACTIC ACID BACTERIA FERMENTATION ON THE PROXIMATE AND MINERAL COMPOSITIONS of Digi taria exilis.” Journal of Experimental Research 12, no. 1 (2024).
55. Ilowefah, Muna, Jamilah Bakar, Hasanah M. Ghazali, Ahmed Mediani, and Kharidah Muhammad. “Physicochemical and functional properties of yeast fermented brown rice flour.” Jour nal of food science and technology 52, no. 9 (2015): 5534-5545.
56. Ongol, Martin Patrick, Eugène Niyonzima, Innocent Gisan ura, and Hilda Vasanthakaalam. “Effect of germination and fermentation on nutrients in maize flour.” (2013): 183-188.
57. Jood, S., N. Khetarpaul, and R. Goyal. “Effect of germi nation and probiotic fermentation on pH, titratable acidi ty, dietary fibre, β-glucan and vitamin content of sorghum based food mixtures.” J. Nutr. Food Sci 2, no. 9 (2012): 1-4.
58. Zhang, Zhenya, Zhongfang Lei, and Ye Chen. “Chemi cal composition and bioactivity changes in stale rice after fermentation with Cordyceps sinensis.” Journal of bio science and bioengineering 106, no. 2 (2008): 188-193.
59. Suarti, Budi, and Slamet Budijanto. “Bio-active compounds, their antioxidant activities, and the physicochemical and pasting properties of both pigmented and non-pigmented fermented de-husked rice flour.” AIMS Agriculture & Food 6, no. 1 (2021).
60. Ejigui, Jeanne, Laurent Savoie, Johanne Marin, and Thérèse Desrosiers. “Beneficial changes and drawbacks of a traditional fermentation process on chemical composi tion and antinutritional factors of yellow maize (Zea mays).” Journal of Biological Sciences 5, no. 5 (2005): 590-596.
61. Tamene, Aynadis, Susanna Kariluoto, Kaleab Baye, and Christèle Humblot. “Quantification of folate in the main steps of traditional processing of tef injera, a cereal based fermented staple food.” Journal of cereal science 87 (2019): 225-230.
62. Doblado, Rosa, Juana Frias, Rosario Muñoz, and Concep ción Vidal-Valverde. “Fermentation of Vigna sinensis var. carilla flours by natural microflora and Lactobacillus species.” Journal of Food Protection 66, no. 12 (2003): 2313-2320.
63. Granito, Marisela, Juana Frias, Rosa Doblado, Marisa Guer ra, Martine Champ, and Concepción Vidal-Valverde. “Nutri tional improvement of beans (Phaseolus vulgaris) by natural fermentation.” European Food Research and Technology 214, no. 3 (2002): 226-231.
64. Barampama, Zacharie, and Ronald E. Simard. “Effects of soaking, cooking and fermentation on composition, in-vitro starch digestibility and nutritive value of common beans.” Plant foods for human nutrition 48, no. 4 (1995): 349-365.
65. Frias, Juana, Martha L. Miranda, Rosa Doblado, and Concepción Vidal-Valverde. “Effect of germination and fermentation on the antioxidant vitamin content and antioxidant capacity of Lupinus albus L. var. Multolupa.” Food Chemistry 92, no. 2 (2005): 211-220.
66. Onwurafor, E. U., J. C. Onweluzo, and A. M. Ezeo ke. “Effect of fermentation methods on chemical and microbial properties of mung bean (Vigna radiata) flour.” Nigerian Food Journal 32, no. 1 (2014): 89-96.
67. Malomo, Adekunbi Adetola, Sumbo Henrietta Abiose, and Hezekiah Adekanmi Adeniran. “Effects of substitution of acha and soybean on alpha-amylase activity, sugars and total free amino acid during production of maize masa.” (2019): 534-538.
68. Osman, Magdi A. “Effect of traditional fermentation pro cess on the nutrient and antinutrient contents of pearl millet during preparation of Lohoh.” Journal of the Sau di Society of Agricultural Sciences 10, no. 1 (2011): 1-6.
69. Abd Elmoneim, O. Elkhalifa, B. Schiffler, and R. Bern hardt. “Effect of fermentation on the functional properties of sorghum flour.” Food Chemistry 92, no. 1 (2005): 1-5.
70. Nkhata, Smith G., Emmanuel Ayua, Elijah H. Kam au, and Jean‐Bosco Shingiro. “Fermentation and ger mination improve nutritional value of cereals and le gumes through activation of endogenous enzymes.”Food science & nutrition 6, no. 8 (2018): 2446-2458.
71. Adeyemo, S. M., and A. A. Onilude. “Enzymatic reduc tion of anti-nutritional factors in fermenting soybeans by Lactobacillus plantarum isolates from fermenting ce reals.” Nigerian Food Journal 31, no. 2 (2013): 84-90.
72. Obadina, A. O., O. J. Akinola, T. A. Shittu, and H. A. Ba kare. “Effect of natural fermentation on the chemi cal and nutritional composition of fermented soymilk nono.” Nigerian Food Journal 31, no. 2 (2013): 91-97.
73. Olagunju, Omotola F., Olufunke O. Ezekiel, Adeni ke O. Ogunshe, Samson A. Oyeyinka, and Oluwato sin A. Ijabadeniyi. “Effects of fermentation on proxi mate composition, mineral profile and antinutrients oftamarind (Tamarindus indica L.) seed in the production of daddawa-type condiment.” LWT 90 (2018): 455-459.
74. Adebiyi, Janet Adeyinka, Eugenie Kayitesi, Oluwafemi Ayodeji Adebo, Rumbidzai Changwa, and Patrick Berka Njobeh. “Food fermentation and mycotoxin detoxification: An African perspective.” Food Control 106 (2019): 106731.
75. Makokha, Anselimo O., Ruth K. Oniang’o, Simon M. Njoroge, and Oliver K. Kamar. “Effect of traditional fermentation and malting on phytic acid and mineral availability from sorghum (Sorghum bicolor) and finger millet (Eleusine coracana) grain varieties grown in Kenya.” Food and nutrition bulletin 23, no. 3_suppl1 (2002): 241-245.