Waghamare Suresh1*, Pandey Akansha1
1Ph.D Scholar, of NIMS University, Jaipur, Rajasthan, India
*Correspondence: Waghamare Suresh, Ph.D Scholar, of NIMS University, Jaipur, Rajasthan, India. E-mail: waghasuresh@gmail.com
Received: 27 Oct, 2025; Accepted: 14 Oct, 2025; Published: 25 Nov, 2025
Citation: Waghamare Suresh, Pandey Akansha. “Peptic Ulcer Disease and the Role of Medicinal Plants in Treatment and Prevention” J Alter Med Ther (2025): 107. DOI: 10.59462/3068-532X.2.1.107.
Copyright: © 2025 Waghamare Suresh. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Background
Up to 10% of people worldwide suffer with peptic ulcer disease, making it a common yet serious chronic condition. Peptic ulcers develop when stomach juice pH is high and mucosal defences are weakened. Both the infection with Helicobacter pylori (H.) and nonsteroidal anti-inflammatory medicines (NSAIDs) have been linked to decreased mucosal resilience to damage. Main body of the abstract Internal gastrointestinal (GI) disruption due to the production of gastric acid or pepsin is what defines peptic ulcer disease (PUD). The stomach and the first part of the duodenum are common sites for the phenomenon. The jejunum, distal duodenum, and lower oesophagus might be affected. Patients with gastric ulcers often have epigastric discomfort 15-30 minutes after eating, whereas those with duodenal ulcers suffer pain 2-3 hours after eating. Short Conclusion Side effects, relapses, and medication interactions have been reported with peptic ulcer therapies such as proton pumps inhibition chemicals and histamine (H2) receptor inhibitor molecules. However, the chemical compounds found in medicinal plants may be used to cure and prevent various illnesses. Therefore, in this analysis, we will look at some of the most often used medicinal plants for peptic ulcers and how they may be put to use in these capacities.
Keywords
Peptic ulcer, gastric disease, H. pylori gastric infection, proton pump inhibition medicine
Introduction
Infected mucosa lining with the defect spreading across the layer beneath the mucosa or propria characterises the occurrence of peptic ulcer infection that develops in the lining of the stomach or proximal duodenum due to the presence of stomach acid. [1]. Although recent investigations involving the occurrence of peptic ulcer have demonstrated a decline in the incidence, hospital admission rates, and death rates related to peptic ulcer [2,3] the condition is still thought to impact 5-10% of the general population. This is probably because of the drop in the number of H. pylori infections that has come from the availability of new medicines and better cleanliness. Historically, it has been thought that an acidic environment with more secretion, in combination with dietary variables or stress, is to blame for mucosal disturbance in individuals with acid peptic illness. Infection with the bacterium Helicobacter pylori, use of alcohol and cigarettes, NSAIDs, and Zollinger-Ellison syndrome are all associated with an increased risk of peptic ulcer [5]. Both infection with Helicobacter pylori and nonsteroidal medicines with anti-inflammatory effects and usage are major negative factors for causing stomach and duodenal ulcers [6]. Susceptibility to an infection is critical in the early stages of mucosal injury, since only a small population shown in the percentage form as patients infected with H. pylori or taking nonsteroidal medicines with anti-inflammatory effects acquire peptic ulcer disease. Peptic ulcers are linked to functional polymorphisms in many cytokine genes. Mucosal interleukin 1 production is altered by IL1B polymorphisms, which in turn contribute to H. pylori associated gastroduodenal disorders [7]. However, peptic ulcer complications are elevated by a factor of four in nonsteroidal infection with anti-inflammation in the users and by a factor of two in aspirin consumers [8].
When anticoagulants, corticosteroids, or selective serotonin reuptake inhibitors are used with nonsteroidal anti-inflammatory medicines or aspirin, upper gastrointestinal bleeding is more likely to occur [9]. Peptic ulcer disease is common in those who use nonsteroidal with anti-inflammation medicines (NSAIDs), however the role of NSAIDs and H. pylori in developing this condition is still up for debate. Peptic ulcer disease risk is increased by nonsteroidal anti-inflammatory drugs (NSAIDs), aspirin usage, and infection with Helicobacter pylori separately, according to a meta-analysis of qualitative research [10]. About 20% of patients with peptic ulcer disease have what is known as an idiopathic ulceration since they test negative for H. pylori, NSAIDs, and aspirin [11]. However, the pathogenic processes underlying the onset of idiopathic peptic ulcers remain unclear [5]. This condition is brought on by an incongruity between elements that support mucosal integrity and inflammatory insults. According to research out of Denmark [12], peptic ulcer rates might rise due to emotional distress. Ischemia, medicines (steroidal medicines, agents used in chemotherapy), radiation, viruses, histamine, which infiltration of eosinophils or the microbes which cause infection during bypass surgery, metabolic abnormalities, and other conditions may also cause this [13].
Mechanism of peptic ulcer infection:
H. pylori are still a leading cause of peptic ulcer disease, affecting about half of the world’s population [14]. African, Central American, Central Asian, and Eastern European nations had a higher H. pylori prevalence [15]. The majority of cases occur in children living in low-income nations with poor sanitation and overcrowding. Through an inflammatory response including lymphocytes, plasma cells, neutrophils, and macrophages, H. pylori induces cells of epithelium degradation and damage, which is often more severe in the antrum. It is unclear how H. pylori trigger the formation of various lesions in the gastric and duodenal mucosa. Peptic ulcer type determines whether H. pylori infection causes hypochlorhydria or hyperchlorhydria. However, H. pylori may also directly
influence the –subunit of hydrogen and potassium ATPase infection, stimulate calcitonin gene-related neurons with sensory nerve endings connected to somatostatin, or limit gastrin synthesis [16]. The major transmitters of the infection caused by H. pylori are cytokine that restrict cell secretion in the parietal region of the stomach in about a range of 10%-15% of the individuals infected with H. pylori infection exhibit increased secretion in the digestive system and a content of somatostatin hormone present in the lower antrum region (Table 01) [17], even though gastric ulcer development is related with hyposecretion. The result is an increase in acid and pepsin production from parietal or gastric cell due to histamine release. When H. pylori are eliminated, gastrin mRNA expression drops but somatostatin mRNA expression rises [18]. Gastric ulcers are linked to mucosal shrinkage in the vast majority of the remaining individuals suffering from peptic ulcer infection. Damage to the gastroduodenal mucosa is caused by nonsteroidal anti-inflammatory drugs (NSAIDs) because of their systematic inhibition of continually expressed (COX-1) called cyclooxygenase, the enzyme responsible for the synthesis of prostaglandin hormone. This results in a reduced mucosal blood movement with a decrease in mucus production and a decrease in the secretion of bicarbonate ions, and an inhibition of response which causes the movement of the ions inside the cell.
Nonsteroidal anti-inflammatory medicines inhibit this enzyme, although the effect is reversible and dose dependent. Using cyclooxygenase-2 (COX-2) selective nonsteroidal medicines with anti-inflammation mechanism and exogenous prostaglandins decrease mucosal damage and ulcer risk [19]. However, NSAIDs’ toxicity varies due to changes in their physicochemical characteristics [20]. NSAIDs initiate damage to the mucosa because they alter mucus phospholipids and cause separation of mitochondrial oxidative phosphorylation. Gastric juice has a pH of 2, thus when NSAIDs are exposed to this acidic environment, they get protonated and are able to pass lipid membranes and enter epithelial cells, which have a pH of 7.4. In this configuration, NSAIDs are unable to penetrate the lipid membrane and accumulate within epithelial cells, where they cause mitochondrial dysfunction, cellular permeability, and a loss of cellular integrity. Large-risk groups for NSAID-induced ulcers include patients with previous instances of the occurrence of ulcers in the gastric region or bleeding, patients who are above 65 years, patients using steroids or anticoagulant medications, and those taking large dosages or a combination of NSAIDs [1].
The diagram showing the mechanism of action of different medicines for treating peptic ulcer is given below as follows (fig 1)
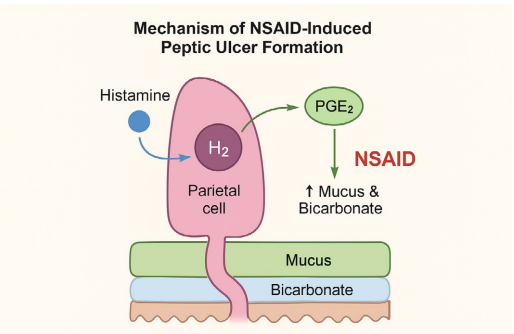
Fig. 1: Shows the action of different peptic ulcer treating medicines in the gastrointestinal system [21]
Inhibitors of Helicobacter pylori:
1. Bismuth
2. Metronidazole
3. Tetracycline
4. Clarithromycin
5. Amoxicillin
| Type of peptic ulcer medicine | Name of medicine | Mechanism of action of peptic ulcer medicine | Side effects of medicines |
| 1.Proton-pump inhibitor medicine | Omeprazole, Lansoprazole, Pantoprazole | The mechanism involves the inhibition of the H+-K+ATPase channel involving the movement of the ions. | Headache, pain in the abdomen, vomiting, vitamin deficiency and flatulence. |
| 2.Histamine receptor inhibiting medicines | Cimetidine, Famotidine, Ranitidine | Inhibits the secretion of histamine in the parietal cells lining the digestive system. | The side or adverse effects are anxiety, depression, and dizziness. |
| 3.Antacid treatment medicines | Use of aluminium hydroxide and magnesium hydroxide powder. | Aluminium hydroxide blocks the action of the pepsin enzyme. | The side effects are vomiting, bad taste of mouth, cramping in the abdomen and imbalance of electrolytes in the body. |
| Magnesium hydroxide causes the retention of osmotic fluid in the body. | |||
| 4.Protective medicines | Misoprostol and Sucralfate | Enhances the secretion of the mucous fluid and augments the development of the lining of the gastrointestinal tract. | The side effects are pain in the head, pain in the body and abdomen pain. |
Table. 1 Shows the name of the peptic ulcer treating medicine, mechanism of action and the side effects of these medicines on the body
Mechanism of action of Vonoprazan for treating peptic ulcer
Pharmacological mode of action with mechanism of action of Vonoprazan medicine
The maximum concentration in the blood (Cmax) of vonoprazan rises from 10 to 60 ng/mL in just 1.5-2 hr,
making it acid-stable and making it possible for it to function as a fast-release treatment. In addition, intestinal meal absorption has a huge impact on its duration of action, with an area calculated in the graph region ranging from in the levels of 0 to beyond scope levels with a dose range of 1.14 to 1.32 ng.hr/mL [22,24-25]. Vonoprazan has a larger beneficial Cmax, AUC, and the half-life compared with PPI, which is but there are no significant variations between the two in terms of retention ratios at pH > 4 and time to achieve Cmax. Since it is found in larger concentrations in the secretion canaliculi that are present in stomach cells in the parietal region in comparison to the levels present in the plasma fluid, and has greater positively charged ions [29], the negative logarithmic of acid solubility constant (pKa) is more than 9.0[22,26-27]. Alpha-1 acid glycoprotein and albumin are required for its dispersal. Vonoprazan, in contrast to PPI, does not need to be activated by acid. To a lesser extent, cytochrome P450 2B6, CYP2C19, CYP2D6, and SULT2A1 are involved in its hepatic metabolism [22].
Since clarithromycin is a potent CYP3A4 inhibitor, it synergises vonoprazan’s pharmacokinetics by decreasing the drug’s metabolism. However, CYP2C19 is predominantly responsible for the metabolism of PPI, and there is a wide range of polymorphisms and metabolic changes that influence proton pump inhibition efficiencies and the activation of drug metabolism [22,30-31]. After learning that H+/K+-ATPase was essential for the last step in gastric acid release, research on suppression of acid drugs took off. Acid activates PPI, a prodrug that forms disulphide bonds to the cysteine in H+/K+-ATPase [32]. After three to five days of therapy, PPI achieves peak acid stability. Researchers have been examining other acid suppressing medications since PPIs are unable to provide a base environment in the stomach. Alternate mechanisms for limiting H+/K+-ATPase activity include decreased concentrations of potassium ions [32]. By interacting with sodium-potassium ATPase pump, P-CAB medicines like vonoprazan function as inhibitor which acts in a reversible manner of competition of potassium ions. Non covalent binding of vonoprazan to H+/K+-ATPase allows it to survive in the acidic stomach secretory canaliculi environment. By slowly dissociating and inhibiting H+/K+- ATPase production over time, vonoprazan raises stomach pH to around 7 in about 4 hours [22,33-37].
Results and Analysis
Effect of Korean Red Ginseng on peptic ulcer
By limiting the transcription of inflammatory response chemicals like IL-8 and 5-LOX mRNA, as well as by decreasing the production of 5(S)-hydroxyeicosatetraenoic acid, an extract of Korean ginseng which is red in colour plays a crucial role in reducing 5-LOX activity produced by H. pylori. As a result, these processes lessen stomach carcinogenesis. The production of 5-hydroxy eicosatetraenoic acid is reduced, and Korean ginseng with red colour has been demonstrated to be helpful in this regard by inhibiting (5-LOX) mRNA of 5-lipo-oxygenase enzyme and the activity of the other enzymes. Similar to how the antioxidant properties of green tea extract may inhibit the activation of TLR-4 in response to lipopolysaccharide from Helicobacter pylori, it may also inhibit the activation of cyclooxygenase-2 (COX-2) and stimulated nitric oxide synthase (iNOS) transcription variables and their target genes. As a result, these blockades amplify the inflammatory factors that cause stomach mucosal lesions [21]. Korean ginseng which is red in colour, has been shown to protect against in vitro cytotoxicity resulting from H. pylori, as reported by Kim et al. [21]. On the other hand, Korean red ginseng was shown to improve eradication levels of H. pylori, decrease stomach inflammation, and protect against reactive damage to DNA and apoptosis in a prior clinical trial [21].
Effects of Allium sativumon controlling peptic ulcer
The major usage of Allium sativum throughout history has been for its therapeutic characteristics, and the advantages to health from garlic have been widely established. The organosulfur compounds in Allium sativum are responsible for its biological effects; these compounds include cysteine with allyl functional group (SAC) sulfoxides and a peptide consisting of glutamate with allyl and-cysteine functional group. Bioactivity may be easily removed from raw Allium sativum. As a result, several types of its extract have been prepared, each with a unique profile of active components, and their effectiveness has been the subject of extensive study [21]. Antioxidant effects, including the removal of free radicals, the blocking of lipoprotein oxidation, and the reduction of the serum glucose stimulation ofanti oxidant enzymes, have been identified as the primary function of Allium sativum extract. In addition, it had an inhibitory tumorigenic action by increasing apoptosis and the activation of arrest and inhibition of the cell cycle mechanism, and it inhibited H. pylori-induced inflammation of the stomach regions in vivo [21]. Growth of H. pylori was inhibited in in vitro studies when the extracts of acetone. Allium sativum was used [21]. This was due to the presence of chemical named allicin and a chemical containing allyl and methyl functional groups with thio and sulfur groups.
Effect of Cistus Laurifolius on peptic ulcer treatment:
Flavonoids play a crucial function in organisms and are mostly responsible for a wide range of biological activities,including antioxidant protection. Because of their scarcity and high price, scientists have discovered a fast method to synthesise polyoxygenated flavones from readily available and cheap flavanones. In order to create a limited flavone with antibacterial action against H. pylori, the methoxylation and bromination technique 30-demethoxysudachitin was used [21].
Cistus laurifolius extract was used in a wide variety of flavonoid studies. Microbial activity with inhibition testing against H. pylori has shown that 3’-sudachitin metabolite containing a deoxymethyl functional group and sudachitin are the most effective metabolites which prevent the growth and proliferation of microbes and thus prove out to be very effective medicinally. Similar research also found that a Cistus laurifolius extract dissolved in organic solvent chloroform and had potent anti-microbial action against Helicobacter pylori. These studies suggest that isolated flavonoids may be utilised as a supplement to the current therapy for infection with Helicobacter pylori [21]. Several isoflavones, with varying degrees of activity against H. pylori, were studied by Li HQ et al. [21]. In this study, the antibacterial activity of many series of metronidazole flavonoid preparations against H. pylori [21] was assessed. One component has been shown to be responsible for the dramatic increase in the secretion levels of Interleukin-8 levels in gastric cells caused by an H. pylori water extraction. However, novel the flavonoid 6, 7, and (2S)- 40,7-dihydroxy-with methyl-flavan functional group have been demonstrated to be highly potent molecules against H. pylori in studies conducted by Nakagawa et al. [21]. Similar results against H. pylori were found in an extract of chloroform of Cistus laurifolius [21] by Ustun et al. Therefore, isolated flavonoids may be utilised as a substance apart from or in addition to the standard therapy for infection with Helicobacter pylori [21].
Effect of Zingiber Officinalis and Zingiber Zerumbet on controlling peptic ulcer infection:
Ginger, or Zingiber officinalis, is a popular spice used in cooking. The plant extract inhibited the development of colon cancer cells, increased DNA synthesis, and induced apoptosis [21], demonstrating its anticancer properties. Further, 6-gingerol is the primary pungent phenolic component of Zingiber officinalis, and it has several pharmacological actions. Prostaglandin E2 (PGE2) suppression is made easier by gingerol-rich preparations of Zingiber officinalis [21]. However, active phenolic substances like gingerol and zingerone significantly inhibit the parietal cell H+, K+ -ATPase. Because of inhibitory action, gingerol and zingerone’s action is crucial for inhibiting the proton pump and decreasing stomach acid output. In addition, it has been shown to prevent H. pylori-related ulcers [21]. Zingiber officinalis has been shown to have a curative effect as an antioxidant that works for stomach ulcers, as reported by Jiang et al. [21]. Zingiber officinalis extract shows fast digestion, so a local therapeutic effect can’t be elicited; many medications indicate a limited transit duration between 2-4 hours in the layers of the stomach; whatever component is soluble will be immediately absorbed; and they reported some limitations of free Zingiber officinalis extracts [21].
Zingiber zerumbet’s zerumbone has been shown to have an important role in the protection of the secretion of gastric juice and action against an induced ulcer in the organ stomach caused due to the addition of ethanol model in rats, according to research by Sidahmed et al. They found that giving rats zerumbone or omeprazole before they developed ulcers considerably reduced the ulcers’ surface areas relative to the ulcers’ control group counterparts. Pre-treatment with 20 mg/kg of omeprazole prevented 76.77 percent of ulcers from forming (p 0.05), whereas pre-treatment with 5 and 10 mg/kg of zerumbone prevented 75.59 and 88.75 percent of ulcers from forming, respectively. However, additional ulcer models were not used to evaluate zerumbone’s gastroprotective processes; hence, additional processes may be involved, and their effect has to be researched and understood [21].
Discussion
The global trend in using herbal supplements has coincided with an increase in reports of adverse occurrences and medication interactions. There may be pharmacokinetic or pharmacodynamics interactions between a herbal supplement and a pharmaceutical. A pharmacokinetic interaction occurs when a herbal supplement and a pharmaceutical medication have the same route of ingestion, distribution, metabolism, or excretion. This results in a shift in the concentration of the pharmaceutical drug in the blood and a modification of the drug’s pharmacologic effect. Antagonising or amplifying the clinical effects of a co-administered medicine is an example of a pharmacodynamic interaction [21]. This kind of interaction does not need a change in the concentration of the co-administered drug. P-gp transporters like digoxin, doxorubicin, rosuvastatin, and verapamil are inhibited by Allium sativum extract. Controlled clinical studies have not validated the suspected interaction between Allium sativum and warfarin despite extensive research. Also, since it prevents platelets from sticking together, it must be administered with care in patients with clotting abnormalities or who are receiving anticoagulant medication. Although preliminary evidence suggests that Zingiber officinalis increases time of blood flow by inhibiting thromboxane synthesis enzyme called as synthetase, this has not yet been verified in a trial study to be performed experimentally. Due to its potential to impede platelet aggregation, Ginkgo biloba may increase the risk of blood flow or bleeding, particularly when used with anticoagulant medications. Ginkgo biloba flavonoids exhibit antiplatelet action [21], however they have no noticeable effect on human blood clotting or platelet function. When used with an NSAID, it might increase the risk of bleeding [21].
Calcium channel ion movement block medicines, several medicines lowering the incidence of hypertension and statin drugs, and some depression lowering medicines may all have their efficacy reduced by the induction of cytochrome P450 3A4 (CYP3A4) by Panax ginseng [21]. Patients with diabetes may benefit from Panax ginseng’s hypoglycaemic properties, but those using phenelzine may have side effects including headache, shaking, and mania if they take the herb simultaneously [21]. Anion movement causing enzyme 1a1 and anion carrying protein 1a12 are important for transporting quinolone ring structure molecules, beta blocking agents, and imatinib medicine, respectively; green tea ingredients and componentshas been proven experimentally to boost the chemical simvastatin chemical in the body [21] or to inhibit these transports of the medicines inside the cells of the body. Cimetidine’s multiple medication interactions stand out among the standard antiulcer treatments. Many medications, including warfarin, phenytoin, diazepam, chlormethiazole, the beta-blocker and lidocaine, have been shown to interact in clinically significant ways, according to studies. Caffeine’s oxidative metabolism in the liver is reduced by cimetidine; therefore, drinking green tea with it may have a greater impact [21]. This is because cimetidine inhibits CYP1A2, the enzyme responsible for this process.
Conclusion
Peptic ulcer disease is still a common clinical issue that affects individuals of all ages. This widespread condition is projected to continue to have a substantial worldwide influence on health-care delivery, medical economics, and patient quality of life as the incidence of peptic ulcer disorder rises with age. Peptic ulcer disease is still a common clinical issue that affects individuals of all ages. It is predicted that peptic ulcer disease, which becomes more frequent as people age, will continue to have a major influence on health-care their delivery, health economics, and patient quality of life on a worldwide scale.
References