Duckshin Park1,2*, Sehrish Shoukat1,2,3, Muhammad Jahanzaib3
1Korea Railroad Research Institute (KRRI), Uiwang-si, 16105, South Korea
2Transportation and Environment Research Centre, Korea Railroad Research Institute
3Korea University of Science and Technology, Daejeon, 34113, South Korea
*Correspondence: Duckshin Park, Korea Railroad Research Institute (KRRI), Uiwang-si, 16105, South Korea. E-mail: dspark@krri.re.kr
Received: 24 Oct, 2025; Accepted: 11 Nov, 2025; Published: 19 Nov, 2025.
Citation: Duckshin Park. “pH-Driven Comparative Analysis of CO2 Absorption Performance in Liquid-Based Hydroxide Absorbents (NaOH, KOH, Ca (OH)2) under Identical Conditions” J Environ Toxicol Res (2025):115. DOI:doi.org/10.59462/3068-3505.2.1.115
Copyright: © 2025 Duckshin Park. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
The rising concentration of atmospheric CO2, largely driven by industrial and energy-related activities, necessitates the development of efficient, scalable, and sustainable capture strategies. CO2 uptake with strong alkaline hydroxides is a promising pathway to scalable and efficient carbon capture, but comparative control performance testing is still needed. In this work, the absorption characteristics of three 3N hydroxide solutions (NaOH, KOH, and Ca (OH)2) for the capture ofCO2 in the two temperature ranges from 25 °C to 50 oC and a descending sequence from 20 °C to 0 °C are studied. Using a laboratory scale setup, adsorption capacity (mol/L), real-time pH change, and a superficially derived absorption efficiency index (AEI) were employed to compare performance under the same operational conditions. Among the tested sorbents, KOH had the highest absorptivity (0.846 mol/L at 0°C), and AEI was the best of them, owing to its better ionic mobility and buffer capacity. These results suggest KOH is ideal for short-term, high capacity capture, whereas Ca (OH)2 offers advantages in pH stability and byproduct utility. This pH-driven analysis offers valuable insights into hydroxide based absorption systems for future low-carbon and decentralized CO2 mitigation technologies.
Keywords
CO2 Mitigation, Absorption, Liquid Hydroxides.
Introduction
CO2 emissions from industrial, transportation, and human activities are major factors contributing to global warming, posing a pressing challenge that is threatening the ecolog ical stability of every species living on planet Earth. Histor ically, scientists have identified the CO2 concentration of 350 parts per million (ppm) as the critical threshold for at-mospheric carbon dioxide [1]. However, this limit has been surpassed, with global atmospheric CO2 concentration reaching 424 ppm in 2024 according to the National Oce anic and Atmospheric Administration [2]. This drastic esca lation is driven by growing energy generation demands to meet the increasing global population requirements. The primary sources of atmospheric CO2 stem from the com bustion of coal, furnace oil, diesel, and natural gas. These industrial processes contribute approximately 65% of global GHG emissions [3]. The Global Carbon Project re port in 2023 states the global industrial emission at 17giga gigatons in 2023-CO2 being released in vast quantities [4] To mitigate a drastic increase in atmospheric CO2, vari ous separation strategies have been developed, including direct air capture, membrane systems, cryogenic sepa ration, adsorption using porous solids, and chemical ab sorption [5]. The carbon dioxide separation methods that are commercially used include CO2 adsorption on a solid catalyst surface, and absorption in chemical solvents and liquids [6], [7]. The adsorption process involves the selective binding of CO2 onto adsorbent material surfac es such as metal oxides, zeolites, metal-organic frame works (MOFs), or activated carbon. Recent advancements in research showed that amine-functionalized adsorbents at ambient conditions have achieved high adsorption ca pacities exceeding 3 mmol/g [8]. The absorption process, on the other hand, implies the process of dissolving CO2 into chemical solvents, typically amines like monoethanol amine (MEA) or hydroxide solutions such as NaOH, Ca (OH)2, and KOH to form stable compounds like carbonates or bicarbonates [7,9]. This method has great applications in industries where absorption systems are required to capture more than 90% of CO2 from the exhaust [2]. Be yond just capturing CO2, it can be re-purposed and used as a resource and valuable product, such as chemicals (urea), synthetic fuels (methanol), or any building material (concrete made from carbonates). It will promote a circular economy as well as prevent it from returning back into the atmosphere [10]. Chemical absorption with amine-based solvents, particu larly monoethanolamine (MEA), is extensively researched and used in large-scale demonstration projects (Rochelle 2012). CO2 capture typically uses aqueous amines inter acting with CO2 at 40oc, resulting in an unstable salt that is energy-intensive and requires significant heat for solvent regeneration of CO2 at nearly 120oc [11,12]. Despite the dominance of amines, strong hydroxide solutions [13]— notably sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca (OH)2)—have long been recognized for their rapid and stable reactions with CO2, forming carbonate and bicarbonate salts. The ab sorption process is advantageous when used for large scale industrial CO2 capture due to its operational flexibil ity and regeneration ability of solvents via heating at high temperature or reducing system pressures.Several previous studies have explored the use of individ ual hydroxide systems under specific operational contexts, but there remains a lack of comparative investigations conducted under standardized conditions. For instance, NaOH has been widely studied for its fast CO2 absorption rate and high alkalinity [14], KOH has been applied in di rect air capture and regenerative systems[15], and Ca (OH)2 has been utilized in mineralization and flue gas treatment [16]. Liquid hydroxide sorbents, despite their use in CO2 cap ture by scrubbing, have been overlooked by amine-based scrubbers. Moreover, pH, despite being a readily mea surable indicator of hydroxide consumption and carbon ate formation, has been underutilized as a real-time diag nostic tool [17]. The primary purpose of this research is to evaluate the absorption rates of CO2 using a selection of liquid-based hydroxides, specifically sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hy droxide (Ca (OH)2), precisely at controlled concentrations across a varying range of temperatures. By quantifying absorption capacities in L/min along with associated ki netics, and pH variations, this study aims to present the first systematic and temperature-resolved comparison of CO2 absorption performance using strong hydroxide solu tions, NaOH, KOH, and Ca (OH)2 solutions, both under heating and cooling conditions under identical conditions. This research shows a pH-driven CO2 absorption analysis, where pH is used as real real-time indicator of hydroxide consumption and carbonate/bicarbonate formation, deter mining the nature of resulting carbonate species affecting absorption capacities and reuse potential.
Experimental details
Materials and Methods
In this study, a series of experiments was conducted to observe the absorption of CO2 in freshly prepared 3N solu tions of NaOH, KOH, and Ca (OH)2 at different tempera tures. To ensure purity and consistency in CO2 absorption experiments, analytical grade chemicals, sodium hydrox ide (NaOH, 99.0%, pure pellets), Potassium hydroxide (KOH, ≥85% pure pellets), and calcium hydroxide (Ca (OH)2, ≥95%, powder) from Sigma-Aldrich (Merck) were used. Deionized water was used to form 3N solutions. Pure carbon dioxide gas from compressed gas cylinders (CO2 purity 99%) from Sigma Aldrich was utilized for this study. A calibrated pH meter with an accuracy of ±0.01 pH units was used to conduct pH measurements. To mini mize the contaminants, acid-washed and deionized rinsed glassware such as beakers, volumetric flasks, and gradu ated cylinders were used for the experiments.
Preparation of 3N NaOH, KOH, and Ca (OH)2 Solution
To absorb CO2, 3N NaOH, KOH, and Ca (OH)2 were fresh ly prepared by dissolving NaOH and KOH pellets in 500 mL of distilled water. Ca (OH)2 was used in powdered form. As NaOH and KOH are monoprotic bases (1N = 1M), it shows that 3N NaOH and KOH solution corresponds to 3 M. For prepar ing the 3N solution of NaOH and KOH, respective pellets were dissolved in 500 mL of distilled water and stirred at 300 rpm with a magnetic stirrer until the pellets were fully dissolved. The initial pH of both NaOH and KOH solutions was adjusted to 13.0 ± 0.1, which aligns with the solution’s expected alkalinity [18]. To ensure the purity and concen tration accuracy of solutions within ±2%, a 10 mL aliquot of each was titrated against 1M HCL. The formation of a 3N solution of NaOH and KOH is an exothermic process, so the mixture was allowed to cool down to room tempera ture. The homogeneity of the mixtures was ensured by the inversion of the flasks several times. Ca (OH)2 due to its limited solubility and diprotic nature for making a true 3N solution presents a unique challenge. A saturated mixture with excess solid was prepared to nor malize 3N equivalence for experiments [5]. The Ca (OH)2 powder was added to 500 mL of deionized water and stirred vigorously at 500 rpm for 30 minutes at room tem perature to ensure maximum dissolution to form a saturat ed solution. The mixture was allowed to settle and filtered by Whatman filter paper (No.1) to remove any undissolved Ca (OH)2 particles from the solution. The initial pH of the solution was ~12.5 ± 0.1, which shows its lower solubili ty than KOH and NaOH. The concentration accuracy was confirmed by 1 M HCL titration.
Experimental Setup for CO2 Absorption
The above-prepared 3N NaOH, KOH, and Ca (OH)2 solu tions were used in a lab-scale setup to assess the CO2 absorption capacities. The experiments were conducted at varying temperatures, and CO2 absorption kinetics were studied under controlled environments. The initial pH of the system was consistently measured at 13, depicting the highly alkaline nature of the absorbent, which is critical for carbonate formation. The temperatures were systemati cally varied in 2 ranges- an ascending sequence: 25 °C to 50oC and a descending sequence from 20 °C to 0oC with a consistent increment and decrement of 5oC. The absor bent volume was fixed at 500 mL, and 30 minutes were provided for each cycle to maintain consistency across all experiments. The schematic of the lab-scale experimental setup used is shown in Fig.1. 500 mL of each represen tative solution (NaOH, KOH, Ca (OH)2) was added into the reactor, leaving half of the vessel empty for maximum liquid-gas phase contact and accommodating potential volume expansion during the CO2 absorption process. CO2 gas was introduced from a cylinder via a mass flow controller. The flow rate of gas was maintained at 1 L/min. The reactor was heated with an external heating system, and a type K thermocouple was used to monitor and verify the temperature inside the reactor. Each temperature was maintained for 10 minutes before CO2 introduction to the reactor to ensure thermal equilibrium. The initial and final pH of the solutions was checked by a pH meter, and a direct electrode was installed directly into the reactor system to monitor the varying pH of the solution during the absorption process.
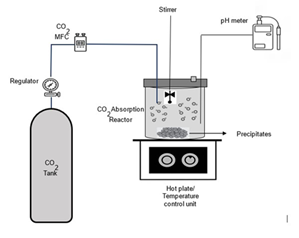
Figure 1: Experimental Setup for CO2 absorption
Results and Discussion
In this study, various solvent types (NaOH, KOH, Ca (OH)2), both under heating and cooling conditions, for CO2 absorption under similar conditions, using pH decay as an indicator across, were used to determine their effect on CO2 absorption. 3N concentration of NaOH, KOH, and Ca (OH)2 was used for this purpose. Each Experimental run was conducted for 30 minutes with a freshly prepared 3N solution for each run. The initial pH of the freshly prepared solution was maintained at 13.
Chemical Absorption mechanism of CO2 is NaOH, KOH, and Ca (OH)2
Chemical Absorption Mechanism of CO2 in NaOH
When NaOH is dissolved in pure deionized water, it com pletely dissociates into Na+ and OH- because of its strong ly alkaline nature. When CO2 in gaseous form is introduced into the solution for the absorption process, it converts into aqueous CO2. Later, CO2 reacts with OH- to form HCO3- and CO3-2 as shown in Eqs. (1), (2).
CO2 (aq) + OH-(aq) > HCO3- (1)
HCO3- (aq) +OH- (aq) > H2O (l) +CO32- (aq) (2)
In a higher pH range, reactions 1 and 2 have very fast kinetics of reversible reactions [19]. The reaction in Eq. (2) dominates the equation. (1) because of the relatively high alkalinity of CO32- than HCO3-, which favors this reaction in even the early stages of the reaction [20]. Therefore, with the increasing CO32- concentration during the initial phase of this phenomenon, the pH of the system is rapidly decreased. The overall pH of the CO2 absorption mechanism reaction in aqueous NaOH can be expressed as eq. (3).
2NaOH (aq) +CO2 (g) → Na2CO3 (aq) + H2O (l) (3)
Na2CO3 produced in the system remains as Na+ and CO32- in the absorbent, while CO2 is continuously absorbed, which favors a forward reaction of eq. (2) causing CO32- accumulation, due to which the pH of the system decreases. The overall reaction during the 2nd range of absorption process can be written as Eq. (4) [19].
Na2CO3- (aq) + CO2 (g) + H2O (l) → 2NaHCO3 (4)
After completing this reaction at equilibrium, the net reac tion of Eqs (3) and (4) can be expressed as Eq. (5), which is the overall CO2 absorption reaction in NaOH solution
NaOH (aq) + CO2 (g) → NaHCO3 (aq) (5)
Chemical Absorption Mechanism of CO2 in KOH
When CO2 is introduced at a higher pH>10 in aqueous KOH solution, the same reaction as in eq. (1) and (2) oc cur. KOH is assumed to be completely dissociated in water by the equation. (6) [21].
KOH (aq) → K+ + OH- (6)
K2CO3 is formed by the reaction of CO32-, formed in pre vious steps, with K+ ions in solution.
The net mechanism of CO2 absorption in KOH can be stat ed as eq. (7)
2KOH (aq) + CO2 (g) → K2CO3 (aq) + H2O (l) (7)
Chemical absorption of CO2 in Ca (OH)2
Ca (OH)2 shows a limited dissolution in water due to its low solubility. A saturated solution formed with excess un dissolved Ca (OH)2 comprises of products shown in eq. (8)
Ca (OH)2 (s) ↔︎ Ca2+ (aq) + 2OH- (aq) (8)
Ca2+ (aq) + CO32- (aq) ↔︎ CaCO3 (s) (9)
CO2 is dissolved in the aqueous phase of Ca (OH)2 solu tion, forming CO2 (aq) as described in equations (1) and (2). The bicarbonate ion (HCO3-) combines with the Ca2+ ions to form insoluble calcium carbonate. The net re action is stated in Eq. (9) [14]. The precipitation of CaCO3 takes place rapidly. This precipitation can also occur on the surface of undissolved Ca (OH)2, leading to hindered dissolution of Ca (OH)2 [22].
Effect of Temperature on CO2 Absorption
Figure 1(a) presents the amount of CO2 absorbed in Li ters by each hydroxide, i.e, NaOH, KOH, and Ca (OH)2, at various temperatures ranging from 25 o C to 50 o C. All three hydroxides show a consistent trend of decreas ing CO2 absorption capacities with increasing tempera ture. This inverse temperature dependence is attributed to the thermodynamic behavior of CO2 in aqueous systems, where gas solubility decreases as temperature increases, which further reduces the absorption capacities of the sys tem [23]. Figure 1(b) shows the CO2 absorbed in Liters by each hydroxide, i.e, NaOH, KOH, and Ca (OH)2, at temperatures ranging from 25 o C to 0 o C.
Among the three hydroxides, 3N KOH exhibits the highest CO2 absorption across the entire temperature range, fol lowed by NaOH. At 25 °C, it absorbed 0.392 mol/L (4.8L) of CO2 at STP, over 30 minutes, which decreased to 0.068 mol/L (0.9L) of CO2 at 50 °C. A similar trend for NaOH is observed, which absorbed 0.343 mol/L (4.2L) CO2 at 25 °C. absorption dropped sharply to 0.038 mol/L (0.5 L) CO2 as temperature rises to 50 °C. The relatively slower de cline rate of KOH suggests that KOH maintains better per formance under thermally stressed conditions, mainly due to higher ionic conductivity and diffusivity of K+ ions [24]
Ca (OH)2 shows the lowest absorption capacity across the entire temperature range. At 25 °C, the solutions absorbed 0.326 mol/L (4L) CO2, decreasing to 0.098 mol/L (1.3L) CO2 at 50 °C, which is expected due to its limited solu bility in water, which restricts the availability of hydroxide ions, and early precipitation of CaCO3, which reduces the gas-liquid reaction [25].
The observed order of efficiency KOH> NaOH> Ca (OH)2 is consistent with prior studies examining individual hy droxides. For instance, [15] in their study found that KOH, when used as an absorbent for gaseous CO2, outperforms NaOH in terms of initial reaction rate and absorption ca pacity, owing to its higher K⁺ ion mobility and similar re-action mechanism, which may enhance the transfer and reaction kinetics during CO2 absorption [24]. On the other hand, the limited performance of Ca (OH)2 is well estab lished in heterogeneous systems, where the formation of CaCO3 precipitate rapidly removes CO3-2 ions from solution, reducing further absorption capacity [26].
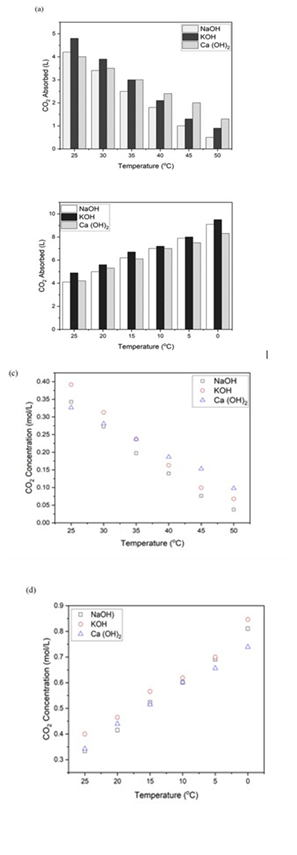
Figure 2: (a) CO2 absorption (L) at 25 °C - 50 °C, (b) CO2 ab sorption (L) at 25°C - 0 °C (c) CO2 absorption (mol/L) at 25 °C - 50 °C, (d) CO2 absorption (mol/L) at 25°C - 0 °C
To further evaluate the thermodynamic absorption per formance of Hydroxide solutions, in addition to total CO2 absorbed, the concentration of absorbed CO2 (mol/L) is also calculated as an indicator of the solution’s CO2 load ing capacity, specifically when the solution used is normal ized by volume. Figure 1 (c) shows the CO2 concentration (mol/L) across temperature 25 °C - 50 °C, and (d) shows the CO2 concentration (mol/L) across temperature 25 °C - 0 °C. These results show a direct comparison of CO2 molar concentrations across different hydroxides under identical thermal conditions.
Final pH as an Indicator of CO2 Absorption
The final pH of each hydroxide solution following 30 min utes of CO2 exposure is shown in Figure 2 (a) for increasing temperature (25 °C - 50 °C) and (b) for decreasing temperature (25 °C - 0 °C). The observed pH provides insights into acid-base reactions between CO2 and OH- ions and the nature of products formed. With the increase in temperature from 25 °C - 50 °C, the final pH of all three hydroxide solutions decreased, which indicates the consumption of OH- ions through CO2 absorption. The final pH of NaOH solution decreased from 12.4 at 25 °C to 10.0 at 50 °C, representing the largest pH drop of 2.4 units. This shows substantial bicarbonate and carbonate formation, consuming free OH- ion as expected from the following reaction:
CO2 + OH- → HCO3-, (10)
HCO3- + OH- → CO3-2 + H2O (11)
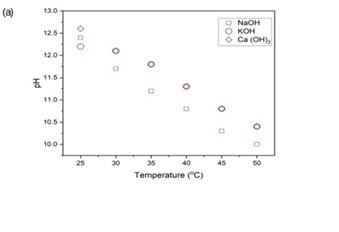
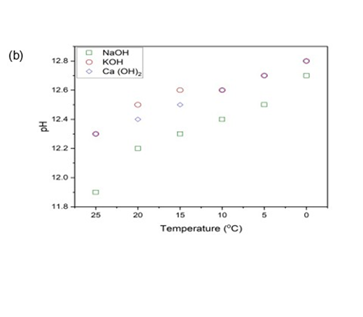
Figure 3: (a) shows pH at 25 °C - 50 °C & (b) shows pH at 25°C - 0 °C
The pH of the KOH solution dropped from 12.2 at 25 °C to 10.4 at 50 °C, slightly less than NaOH, confirming a similar reaction pathway of OH- neutralization. Although both NaOH and KOH are strong bases and follow similar carbonate formation pathways, the pH decline during CO2 absorption was slightly less in KOH (∆pH 1.8) compared to NaOH (∆pH 2.4). This marginal difference in the final pH of NaOH and KOH solutions can be due to the higher ionic mobility of K+ ions and weaker hydration shell compared to Na+ ions, which leads to faster mass transport in solutions, providing more efficient buffering capacity over the duration of the absorption experiment. [27]. Ca (OH)2 showed the pH drop from 12.4 at 25 °C to 10.4 at 50 °C, due to its precipitation of CaCO3, which limits further pH drop by removing CO32- ions from solution. This buffering behaviour is consistent with previous studies [25], which reported pH stabilization in calcium-based systems due to heterogeneous CaCO3 formation. This decreasing trend with increasing temperature supports the inverse relation ship between gas solubility and temperature, indicating less CO2 is absorbed at elevated temperatures [23]. In the reverse temperature regime, i.e., 25°C - 0 °C (Fig. 2(b)), the pH of all three hydroxides increased, confirming that more CO2 is absorbed at lower temperatures due to higher solubility of gases at lower temperatures. For NaOH and KOH, pH rose from 11.9 to 12.7 and 12.3 to 12.8 at 25 °C- 0 °C, respectively, depicting the role of cooling in suppressing bicarbonate formation and preserving alkalinity. Ca (OH)2, starting at pH 12.3 at 25 °C, increased to 12.8 at 0 °C, consistent with minimal ionic conversion due to CaCO3 precipitation dominating the equilibrium. These results support the idea that at lower temperatures, fewer OH- ions are consumed per mole of ab sorbed CO2, because the carbonate pathway may shift towards physical stability rather than chemical neutraliza tion.
Absorption Efficiency Index
To further compare the performance of the three hydroxide solutions under cooling conditions (25 °C- 0 °C), we pro pose the absorption efficiency index (AEI) at each interval. This index shows the moles of CO2 absorbed per unit de crease in alkalinity, showing how efficiently each hydroxide
converts OH- to carbonates without excessive pH loss. AEI is calculated by the following equation (12): AEI = CO2 absorbed (mol) / ∆ pH (12) Where:
CO2 absorbed (mol) = total moles of CO2 taken up over 30 minutes
∆ pH = Initial pH – Final pH
*Since the initial pH for all solutions is 13, we can compare each case.
| Hydroxide | Temperature | Total CO2 absorbed (moles) (30 minutes) |
∆ pH | |
| (°C) | ||||
| NaOH | 25 | 0.17 | 1.1 | |
| 20 | 0.21 | 0.8 | ||
| 15 | 0.26 | 0.7 | ||
| 10 | 0.3 | 0.6 | ||
| 5 | 0.35 | 0.5 | ||
| 0 | 0.41 | 0.3 | ||
| KOH | 25 | 0.2 | 0.7 | |
| 20 | 0.23 | 0.5 | ||
| 15 | 0.28 | 0.4 | ||
| 10 | 0.31 | 0.4 | ||
| 5 | 0.35 | 0.3 | ||
| 0 | 0.42 | 0.2 | ||
| Ca (OH)2 | 25 | 0.17 | 0.7 | |
| 20 | 0.22 | 0.6 | ||
| 15 | 0.26 | 0.5 | ||
| 10 | 0.3 | 0.4 | ||
| 5 | 0.33 | 0.3 | ||
| 0 | 0.37 | 0.2 |
Table 1: Total CO2 absorbed at each interval for each 30-min ute experimental run and change in pH drop from 25 °C to 0 °C
| Temperature (°C) | AEI NaOH(Mol/pH unit) |
AEI KOH(Mol/pH unit) |
AEI Ca (OH)2 (Mol/pH unit) |
| 25 | 0.152 | 0.286 | 0.245 |
| 20 | 0.259 | 0.465 | 0.367 |
| 15 | 0.374 | 0.707 | 0.515 |
| 10 | 0.501 | 0.773 | 0.752 |
| 5 | 0.691 | 1.166 | 1.093 |
Table 2: Absorption Efficiency Index (AEI) of each Hydroxide
To more precisely evaluate the relative performance of each hydroxide, Table 2 shows the AEI of each hydroxide absorbent under decreasing temperature conditions. The efficiency index was calculated at each 5 °C interval from 25 °C to 0 °C. KOH demonstrated the highest AEI at all temperatures, rising from 0.286 (Mol/pH unit) at 25 °C to 2.115(Mol/pH unit) at 0 °C. This reflects its ability to absorb CO2 while preserving alkalinity more effectively than NaOH or Ca (OH)2. The higher AEI index of KOH again shows the higher K+ ionic mobility and its favorable effect on buffering and mass transport in solution [24]. Although Ca (OH)2 consistently absorbs less CO2 in absolute terms as compared to NaOH, its AEI is higher at lower temperatures. At 0 °C, the AEI of Ca (OH)2 reaches 1.85 mol/pH unit, which is higher than NAOH’s AEI of 1.35 mol/pH unit. This difference can be because AEI considers not only the quantity of CO2 ab sorbed but also the extent of alkalinity loss, which is mea sured by ∆ pH. When CO2 is absorbed in NaOH, it reacts homogeneously in solution, due to which bicarbonates (HCO3-) and carbonates (CO32-) remain in the aqueous phase, leading to a larger pH decline as OH- ions are con sumed. In contrast, Ca (OH)2 reacts heterogeneously so that CaCO3 precipitates upon CO2 absorption. This sol id-liquid separation buffers the system, leading to a stabi lized pH while still capturing CO2. This ability of CaCO3 to consume CO32- ions from solution limits further acidifica tion and reduces ∆ pH per mole of CO2 absorbed, there fore increasing the AEI. These results show the value of AEI as a comparative metric that captures both chemical efficiency and pH economy in evaluating CO2 scrubbing agents. Similar behaviors have been observed in previous studies of Ca (OH)2 based mineralization and aqueous carbonation [25].
Byproduct Formation and Reusability
Liquid CO2 absorption systems are significantly influenced by the nature, reusability, and economic viability of byprod ucts formed during the process. When NaOH reacts with CO2, it forms sodium carbonate (Na2CO3) or sodium bi carbonate (NaHCO3) in aqueous solutions. Regeneration of NaOH from these carbonates requires high-tempera ture calcination, which is an energyintensive process and poses environmental challenges [28].
Similarly, KOH reacts with CO2 to form potassium carbon ate (K2CO3). The regeneration from K2CO3 also requires high energy input, often involving thermal decomposition or electrochemical methods. [29]
Contrarily, Ca (OH)2, when it reacts with CO2, forms precip itates of CaCO3. This byproduct is stable as well as easily separable and has a high commercial value in industries such as construction, paper, and plastics. Although regen eration from CaCO3 also requires calcination at high tem peratures, direct use of CaCO3 as a byproduct offers more economic benefits for CO2 utilization without the immedi ate need for regeneration [30].
Therefore, although NaOH and KOH offer higher CO2 absorption capacities, Ca (OH)2 presents an advantage in terms of byproduct management, by forming a usable solid precipitate with economic benefits and less energy requirements.
Conclusions and Recommendations This study systematically compared the CO2 absorption characteristics of 3N NaOH, KOH, and Ca (OH)2 under similar conditions in solutions over a wide range of tem peratures. The results revealed that temperature plays a critical role in determining both the absorption capacity and chemical efficiency of hydroxide.
Amongst the three hydroxides, KOH demonstrated the highest CO2 absorption capacity (0.846 mol @ 0 °C) and the highest AEI, reflecting its superior performance in both capacity and pH retention due to its ionic mobility. At room temperatures (25 °C), all three hydroxides demonstrated absorption capacities. KOH exhibited the highest absorption capacity of ~ 0.392 mol/L CO2 with a final pH of ~12, resulting in the highest AEI (0.20 mol/pH unit). NaOH fol lowed by ~0.33 moles/L and a slightly lower final pH of 11.9, yielding an AEI of 0.15 mol/pH unit. Ca (OH)2, despite its lower reactivity and solubility, absorbed ~ 0.32 mol/L of CO2 with a minimal pH loss. These results show that while KOH is the most effective absorber at ambient conditions, Ca (OH)2 shows superior pH stability, and NaOH offers abalanced performance.
For maximum absorption performance and short-term capture or flue gas treatment, KOH is recommended due to its superior CO2 absorption capacity. For the applica tions concerning material reusability and product value, e.g, mineralization, Ca (OH)2 offers advantages despite lower reaction rates.
Future work should explore the byproduct characteristics under identical conditions. Also, technoeconomic assess ment and regeneration strategies for each hydroxide un der identical conditions should be studied, especially in passive, low-temperature, or decentralized CO2 capture systems.
Author Contribution
Conceptualization, Data curation, & Formal Analysis, Seh rish Shoukat, Formal analysis, Muhammad Jahanzaib, Funding acquisition, Duckshin Park; Methodology, Sehrish Shoukat, Muhammad Jahanzaib; Project administration, Duckshin Park; Supervision, Duckshin Park; Writing – original draft, Sehrish Shoukat; Writing – review & editing, Sehrish Shou kat, Muhammad Jahanzaib, and Duckshin Park.
Funding source
This research was funded under the Project “Development of mobile direct carbon dioxide capture technology”, hav ing project code (PK2502C2) under the Ministry of Science and ICT for the Projects, South Korea.
Data availability statement
Data will be made available upon request
Declaration of competing interest
The authors certify that they have no known financial or interpersonal conflicts that might have influenced the re search presented in this study.
Acknowledgment
This research was funded under the Project “Development of mobile direct carbon dioxide capture technology,” hav ing project code (PK2502C2) under the Ministry of Sci ence and ICT for the Projects, South Korea, supervised by Dr. Duckshin Park, and also the National University of Sciences and Technology, Islamabad, Pakistan. We would also like to acknowledge all the volunteers who participat ed in this study
References