Ganiyu M.A1, Karigidi K.O2, Oguntimehin I.I3*
1,2,3Department of Chemical Sciences, School of Science, Olusegun Agagu University of Science and Technology, Okitipupa, Ondo State, Nigeria
*Correspondence: Oguntimehin I.I, Male, Depart ment of Chemical Sciences, School of Science, Olusegun Agagu University of Science and Technology, Okitipupa, Ondo State, Nigeria. E-mail: ii.oguntimehin@oaustech.edu.ng
Received: 13 Oct, 2025; Accepted: 24 Oct, 2025; Published: 31 Oct, 2025.
Citation: Ganiyu M.A, Karigidi K.O, Oguntimehin I.I. “Physiological and Biochemical Responses of Cucumber (Cucumis sativus L.) to Phenanthrene Exposure in an Organically Fertilized Soil.” J Environ Toxicol Res (2025):112. DOI:doi.org/10.59462/3068-3505.2.1.112
Copyright: © 2025 Oguntimehin I.I. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Polycyclic aromatic hydrocarbons (PAHs) are persistent organic pollutants that pose significant threats to agricultural productivity and food safety. This study investigated the physiological and biochemical responses of cucumber (Cucumis sativus L.) cultivated in organically fertilized soil and exposed to phenanthrene (PHE) at concentrations of 0, 5, 10, 15, and 20 μM over 28 days. A completely randomized design with six replicates per treatment was employed. Chlorophyll a and b contents declined significantly (p < 0.05) with increasing PHE concentrations, decreasing by 23% and 28%, respectively, at 20 μM compared to controls. Total phenolic content increased significantly from 52.98 ± 10.99 to 69.16 ± 8.45 mg GAE/g dry weight, while flavonoid content rose from 24.57 ± 5.40 to 32.55 ± 4.46 mg QUE/g dry weight at the highest PHE concentration.
Total antioxidant capacity increased by 25.6% at 20 μM PHE, indicating activation of defense mechanisms. However, radical scavenging activities (DPPH and ABTS) declined significantly, suggesting oxidative stress. Nitric oxide levels increased 3.3-fold at 20 μM PHE, confirming stress signaling responses. Despite biochemical perturbations, fruit yield showed no statistically significant reduction across treatments. These findings demonstrate that cucumber can partially tolerate PHE stress through enhanced antioxidant responses, with organic fertilization providing some protection. However, sustained exposure may compromise crop quality and food safety, necessitating integrated soil remediation strategies.
Keywords
Polycyclic Aromatic Hydrocarbons, Phenanthrene, Oxidative Stress, Antioxidant Defense, Cucumis Sativus, Organic Fertilization, Food Security
Introduction
Polycyclic Aromatic Hydrocarbons (PAHs) represent a major class of persistent organic pollutants generated primarily through incomplete combustion of organic materials, industrial processes, and fossil fuel utilization [1]. Their environmental persistence, combined with semivolatile properties and bioaccumulation potential, makes PAHs a significant concern for ecosystem health and agricultural sustainability [2]. Among the 16 priority PAHs identified by the US Environmental Protection Agency, phenanthrene (PHE), a three-ring PAH, is among the most abundant in agricultural soils and serves as a representative compound for studying plant-PAH interactions [3,4]. Previous research has demonstrated that PAH exposure induces multiple stress responses in plants, including disruption of photosynthetic machinery, generation of reactive oxygen species (ROS), and perturbation of cellular membrane integrity [5]. Plants respond to such Oguntimehin I Istress through activation of antioxidant defense systems, including enzymatic antioxidants (catalase, peroxidase, superoxide dismutase) and non-enzymatic compounds such as phenolic acids, flavonoids, and ascorbic acid [6]. However, these adaptive responses vary considerably among species and may be insufficient to maintain normal growth and productivity under prolonged exposure.
Organic soil amendments have gained attention as sustainable approaches to enhance plant stress tolerance while improving soil fertility. Organic fertilizers, including composted materials, biochar, and waste-derived inputs, can enhance soil structure, water retention, and nutrient availability [7]. Additionally, organic amendments may reduce PAH bioavailability through sorption mechanisms and stimulate microbial degradation processes [8].
Cucumber (Cucumis sativus L.) is an economically important vegetable crop worldwide, valued for its nutritional content and economic significance. However, limited research has examined the physiological responses of cucumber to PAH contamination, particularly under organic fertilization regimes. Understanding these interactions is crucial for developing sustainable agricultural practices in contaminated environments and ensuring food safety.
This study aimed to: (1) evaluate the effects of phenanthrene exposure on chlorophyll content, antioxidant enzyme activities, and secondary metabolite production in cucumber; (2) assess the protective role of organic fertilization in mitigating PHE toxicity; and (3) determine the implications for crop yield and food quality. The findings will contribute to our understanding of plant pollutant interactions and inform strategies for sustainable crop production in contaminated soils.
Materials and Methods
Experimental Site and Plant Material
The experiment was conducted in a controlled screenhouse facility at Olusegun Agagu University of Science and Technology (OAUSTECH), Okitipupa, Ondo State, Nigeria (6°30’N, 4°47’E) during the 2024 growing season. The screenhouse was equipped with UV-resistant polyethylene covering and polyester netting (40-mesh) to minimize pest intrusion while maintaining adequate light transmission (≥85% photosynthetically active radiation). Seeds of cucumber (Cucumis sativus L., cultivar ‘Padma 108F1’) were obtained from the International Institute of Tropical Agriculture (IITA), Ibadan, Nigeria. This cultivar was selected for its uniform growth characteristics and commercial importance in the region.
Soil Preparation and Characterization
Sandy loam soil was collected from the university research farm (0-20 cm depth), air-dried, and sieved through a 2-mm mesh. Soil chemical properties were analyzed following standard procedures: pH (1:2.5 soil:water ratio using pH meter), organic carbon (Walkley-Black method), total nitrogen (Kjeldahl method), available phosphorus (Bray-1 method), and exchangeable cations (1 M NH4OAc extraction). Soil characteristics were: pH 6.2, organic carbon 1.8%, total nitrogen 0.14%, available P 12.5 mg/ kg, and CEC 8.4 cmol/kg.
Organic Fertilizer Preparation and Application
The organic fertilizer was prepared by combining charcoal (500 g), wood ash (500 g), and human urine (1 L), following protocols adapted from [9]. The mixture was sun-dried for 48 hours and ground to pass through a 0.5-mm sieve. Chemical analysis revealed: N (3.2%), P2O5 (2.1%), K2O (4.8%), and organic matter (68.4%). Each plant received 100 g of organic fertilizer incorporated into the soil two weeks post-germination.
Experimental Design and Treatments
A completely randomized design (CRD) was employed with six treatments and six replicates (n=36 plants total). Treatments consisted of:
• T1 (Blank): Deionized water
• T2 (Control): Ethanol vehicle control (0.1% v/v in deionized water)
• T3: 5 μM phenanthrene
• T4: 10 μM phenanthrene
• T5: 15 μM phenanthrene
• T6: 20 μM phenanthrene
Phenanthrene Preparation and Application
Phenanthrene (≥98% purity, Sigma-Aldrich) stock solutions were prepared in ethanol and diluted with deionized water to achieve final concentrations of 5-20 μM. The ethanol concentration in all treatments was maintained at 0.1% v/v to minimize solvent effects. Solutions were prepared fresh daily and stored in amber glass vials at 4°C.
Four weeks after sowing, PHE solutions (10 mL per plant) were applied to soil surface every 48 hours for 14 days (7 applications total). This application method simulates atmospheric deposition and soil contamination scenarios.
Growth Monitoring and Environmental Conditions
Environmental parameters were monitored continuously using digital data loggers (Onset HOBO U23 Pro v2): temperature (24.1 ± 0.8°C), relative humidity (53.7 ± 4.2%), and photosynthetically active radiation (average 400 μmol m-² s-¹ during daylight hours). Plants were irrigated daily with deionized water to maintain soil moisture at 70% field capacity.
Chlorophyll Content Analysis
Chlorophyll extraction was performed at 14 days and 28 days after treatment initiation using the method of [10]. Fresh leaf samples (0.5 g) were homogenized in 10 mL of 80% acetone and extracted in darkness for 24 hours at 4°C. After centrifugation (3000 × g, 10 min), absorbance was measured at 663 nm (Chl a) and 647 nm (Chl b) using a UV-visible spectrophotometer (Shimadzu UV-1800). Chlorophyll concentrations were calculated using standard equations and expressed as mg g-¹ fresh weight.
Chlorophyll a (mg/g) = 13.36 * A664 − 5.19 * A649 (1)
Chlorophyll b (mg/g) = 27.43 * A649 − 8.12 * A664 (2)
Biochemical Analyses
Leaf and fruit samples were harvested at 28 days, immediately frozen in liquid nitrogen, and stored at -80°C until analysis. Samples (1.0 g) were homogenized in 10 mL phosphate buffer (50 mM, pH 7.0) containing 1% polyvinylpyrrolidone and centrifuged (12,000 × g, 20min, 4°C). The supernatant was used for all biochemical assays.
Total Phenolic Content (TPC): Determined using the Folin-Ciocalteu method with gallic acid as standard [11]. Results expressed as mg gallic acid equivalents (GAE)/g dry weight.
Total Flavonoid Content (TFC): Measured using the aluminum chloride colorimetric method with quercetin as standard [12]. Results expressed as mg quercetin equivalents (QUE)/g dry weight.
Total Antioxidant Capacity (TAC): Assessed using the phosphomolybdenum method [13]. Results expressed as mg ascorbic acid equivalents (AAE)/g dry weight.
Lipid Peroxidation: Estimated as malondialdehyde (MDA) content using the thiobarbituric acid method [14]. Results expressed as μmol MDA/g fresh weight.
Radical Scavenging Activity: DPPH and ABTS radical scavenging activities were determined according to [15] and [16], respectively. Results expressed as percentage inhibition.
Nitric Oxide Content: Measured using the Griess reagent method [17]. Results expressed as μmol NO/g fresh weight.
Yield Assessment
Mature fruits were harvested at 90 days after sowing and immediately weighed using an analytical balance (±0.01 g precision). Only fruits reaching commercial maturity (uniform green color, firm texture) were included in yield calculations.
Statistical Analysis
Data were analyzed using R software (version 4.3.0). Normality and homogeneity of variance were assessed using Shapiro-Wilk and Levene’s tests, respectively. One way analysis of variance (ANOVA) was performed for each parameter, followed by Tukey’s HSD post-hoc test for multiple comparisons (α = 0.05). Correlation analysis was conducted to examine relationships between variables. Data are presented as mean ± standard error (SE).
Results
Effects on Chlorophyll Content
Chlorophyll a and b contents showed similar response patterns to PHE exposure (Figure 1). Both pigments increased slightly from day 14 to day 28 in control treatments, but declined significantly under PHE stress. At 28 days, chlorophyll a content decreased from 2.84 ± 0.12 mg/g FW in controls to 2.18 ± 0.15 mg/g FW at 20 μM PHE (p < 0.05). Similarly, chlorophyll b declined from 1.92 ± 0.08 to 1.38 ± 0.11 mg/g FW. The total chlorophyll content showed a dose-dependent reduction, with the most severe decline (28.4%) observed at the highest PHE concentration.
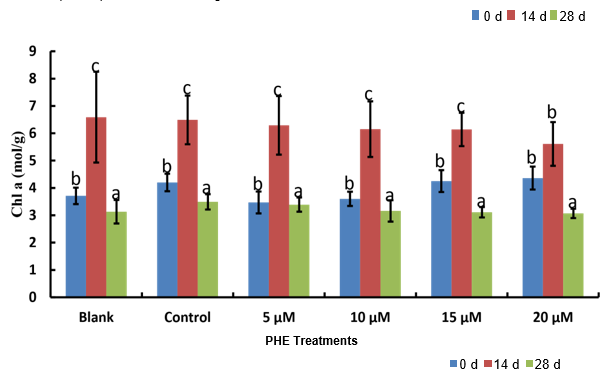
Figure 1: Chl a contents of cucumber plants before, during and after PHE exposure in 2024.). 0d indicates 0 day, 14d indicates 14 days and 28 d indicates 28 days
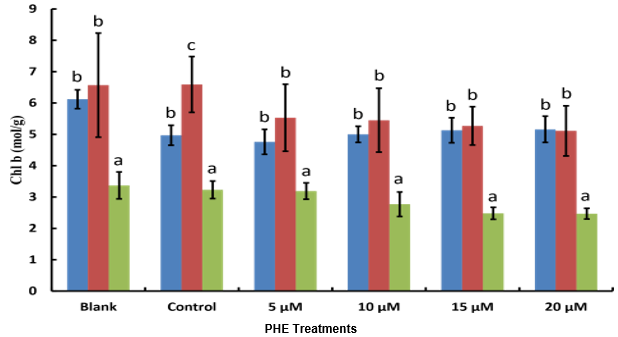
Figure 2: Chl b contents of cucumber plants before, during and after PHE exposure in 2024. Values shown in the figure above are means of six plants measurement in each treatment group ± standard errors. Different letters on each bar indicate significant differences for a PHE Treatments.
Biochemical Responses in Leaves
Total phenolic content increased significantly with PHE concentration, rising from 52.98 ± 10.99 mg GAE/g DW in blanks to 69.16 ± 8.45 mg GAE/g DW at 20 μM PHE (Table 1). This represented a 30.5% increase compared to controls. Flavonoid content showed a similar but less pronounced increase, reaching 32.55 ± 4.46 mg QUE/g DW at the highest PHE level.
Total antioxidant capacity exhibited a biphasic response, initially declining at 5 μM PHE before increasing at higher concentrations. At 20 μM PHE, TAC was 25.6% higher than control values (p < 0.05). Conversely, radical scavenging activities declined under PHE stress. DPPH scavenging decreased from 18.88 ± 3.60% in blanks to 10.25 ± 5.20% at 20 μM PHE, while ABTS activity remained relatively stable across treatments.Nitric oxide accumulation increased significantly with PHE exposure, reaching 8.59 ± 2.89 μmol NO/mg protein at 20 μM PHE compared to 2.58 ± 0.54 in blanks (p < 0.05). Lipid peroxidation showed a slight but non-significant increase across treatments.
| Plant | Parameters | Blank | Control | 5 µM | 10 µM | 15µM | 20 µM |
| Cucumber | Phenolics(mgGAE/g) | 52.98±10.99c | 57.45±8.90bc | 66.93±5.08abc | 67.93±9.58ab | 68.21±3.87ab | 69.16±8.45a |
| Total flavonoids | 24.57±5.40a | 25.19±5.16a | 27.16±3.64a | 28.40±6.85a | 30.68±3.24a | 32.55±4.46a | |
| (mgQUE/g) | |||||||
| TAC | 27.76±4.17b | 31.20±10.09a | 12.36±5.07a | 34.42±4.26a | 34.72±1.48a | 34.85± 3.16a | |
| (mgAAE/g) | |||||||
| LPO (mgAAE/100g) | 0.54±0.04a | 0.55±0.04a | 0.61±0.15a | 0.75±0.20a | 0.76±0.18a | 0.79±0.33a | |
| ABTS (%Inhibition) | 184.0±1.52a | 183.84±1.27a | 183.19±1.32a | 182.14±3.23a | 182.14±2.44a | 181.46±4.94a | |
| DPPH (%Inhibition) | 18.88±3.60a | 17.67±3.02ab | 14.98±3.61abc | 12.38±4.54abc | 11.38±5.14bc | 10.25±5.20c | |
| Nitric Oxide (µmoleNO formed/mgprotein) | 2.58±0.54b | 4.77±1.88ab | 5.09±2.29ab | 6.85±2.55a | 7.39±1.66a | 8.59±2.89a | |
| Values are means of seven determinations from Cucumber plants± standard error (S.E.). Different letters indicate significant difference at P < 0.05 (Tukey’s post-hoc test) | |||||||
Table 1: Effects of PHE Concentration on Biochemical Parameters in Cucumber leaves.
Biochemical Responses in Fruits
Cucumber fruits exhibited more pronounced biochemical changes than leaves (Table 2). Total phenolic content increased dramatically from 16.66 ± 1.96 mg GAE/g DW in blanks to 29.89 ± 3.47 mg GAE/g DW at 20 μM PHE (79.4% increase). Flavonoid accumulation was even more striking, increasing from 5.91 ± 3.26 to 51.94 ± 7.15 mg QUE/g DW (779% increase).
Total antioxidant capacity in fruits increased 2.8-fold at the highest PHE concentration. However, both DPPH and ABTS scavenging activities declined significantly, with DPPH activity decreasing by 54% at 20 μM PHE. Lipid peroxidation increased significantly in fruits, reaching 0.96 ± 0.02 mg AAE/100g at 20 μM PHE compared to 0.54 ± 0.02 in controls.
| Plant | Parameters | Blank | Control | 5 µM | 10 µM | 15µM | 20 µM |
| Cucumber | Total Phenolics (mgGAE/g) | 16.66± 1.96c | 18.94±0.83bc | 21.61±0.83bc | 23.96±0.83ab | 25.72±0.60ab | 29.89±3.47a |
| Total flavonoids (mgQUE/g) | 5.91±3.26c | 12.68±5.61c | 21.85±1.88bc | 25.04±1.76ab | 32.42±6.48ab | 51.94± 7.15a | |
| TAC (mgAAE/g) | 12.78±0.91c | 14.10± 0.56bc | 16.68±2.46bc | 22.30±4.45abc | 28.91±5.30ab | 35.87±7.11a | |
| LPO (mgAAE/100g) | 0.54±0.02c | 0.57±0.05c | 0.70±0.10bc | 0.85±0.03ab | 0.89±0.01ab | 0.96±0.02a | |
| ABTS (%Inhibition) | 190.44±0.29a | 187.73±0.18a | 180.37±1.34b | 176.73±1.53b | 172.09±0.58c | 170.19±1.38c | |
| DPPH (%Inhibition) | 78.88.±0.66b | 76.96±1.10b | 74.92±1.91a | 66.83±7.46a | 50.39±4.87b | 36.09±3.83b | |
| Nitric Oxide (µmoleNO formed/mgprotein) | 2.79±0.89b | 4.21±0.79ab | 5.16±0.08a | 5.34±0.21a | 5.52±0.12a | 5.81±0.05a | |
| Values are means of seven determinations from Cucumber fruit7 standard error (S.E.). Different letters indicate significant difference at P < 0.05 (Tukey’s post-hoc test) | |||||||
Table 2: Effects of PHE Concentration on Biochemical Parameters in Cucumber fruit exposed to phenanthrene.
Fruit Yield
Despite significant biochemical changes, cucumber fruit weight showed no statistically significant differences among treatments (Table 3). Average fruit weights ranged
from 312.45 ± 101.51 g (10 μM PHE) to 369.98 ± 102.34 g (control), with coefficients of variation between 25-30% indicating high variability within treatments.
| Fruit Weight | ||||||
| Plant | Parameter | Blank | Control | 5 µM | 10 µM | 15µM |
| Cucumber | Fruit weight | 462.30± | 369.98± | 329.73± | 312.45± | 320.58± |
| (g) | 112.12a | 102.34a | 63.98a | 101.51a | 81.84a | |
| Values are means of one determination from Cucumber fruit weight± standard error (S.E.). Different letters indicate significant difference at P < 0.05 (Tukey’s post-hoc test). | ||||||
Table 3: Fruit weight in cucumber plants exposed to different concentrations of phenanthrene (PHE)
Correlation Analysis
Correlation analysis revealed significant positive relationships between PHE concentration and phenolic content (r = 0.78, p < 0.001), flavonoid content (r = 0.65, p < 0.01), and nitric oxide levels (r = 0.82, p < 0.001). Negative correlations were observed between PHE concentration and chlorophyll content (r = -0.71, p < 0.001) and DPPH scavenging activity (r = -0.58, p < 0.01).
Discussion
PHE-Induced Photosynthetic Impairment
The significant reduction in chlorophyll content with increasing PHE concentration confirms that PAHs disrupt photosynthetic processes in cucumber plants. This decline likely results from multiple mechanisms, including direct chlorophyll degradation, impaired chlorophyll biosynthesis, and structural damage to chloroplasts [3]. The dose dependent response observed in this study aligns with previous findings in other crop species exposed to PAHs [18].
The organic fertilization appeared to provide some protection against chlorophyll loss at lower PHE concentrations, possibly through enhanced nutrient availability and improved soil structure. However, this protective effect was overwhelmed at higher exposure levels, suggesting that organic amendments alone cannot fully mitigate severe PAH stress.
Activation of Antioxidant Defense Systems
The significant increases in phenolic and flavonoid contents represent a classical plant stress response to environmental pollutants. These secondary metabolites serve multiple protective functions, including ROS scavenging, membrane stabilization, and metal chelation [19]. The greater accumulation in fruits compared to leaves suggests tissue-specific responses, possibly reflecting differential sensitivity or metabolic capacity.
The biphasic response of total antioxidant capacity, with initial decline followed by increase, indicates a complex adaptive process. The initial reduction may reflect consumption of existing antioxidants, while subsequent increases suggest de novo synthesis of protective compounds. This pattern has been observed in other plant-pollutant interaction studies [20].
Oxidative Stress and Signaling Responses
The decline in radical scavenging activities despite increased antioxidant metabolites suggests that ROS production exceeded the capacity of defense systems, resulting in net oxidative stress. This interpretation is supported by increased lipid peroxidation in fruits, indicating membrane damage from oxidative processes.
The substantial increase in nitric oxide levels provides evidence for stress signaling responses. NO serves as a key signaling molecule in plant stress responses, regulating antioxidant enzyme expression and coordinating cellular defense mechanisms [21]. The 3.3-fold increase observed at 20 μM PHE suggests active stress perception and signal transduction.
Crop Yield and Food Safety Implications
The lack of significant yield reduction despite biochemical perturbations indicates partial tolerance of cucumber to PHE stress. This resilience may result from effective compartmentalization of PHE in non-essential tissues or enhanced antioxidant defenses provided by organic fertilization. However, the substantial changes in fruit biochemistry raise concerns about food quality and safety.
The dramatic increases in phenolic and flavonoid contents in fruits could affect nutritional quality, taste, and shelf life. While these compounds have beneficial antioxidant properties, excessive levels may impart bitter flavors or interact with other nutrients. Furthermore, potential accumulation of PAH metabolites in edible tissues warrants investigation from a food safety perspective.
Role of Organic Fertilization
The organic fertilizer used in this study appeared to provide moderate protection against PHE toxicity, evidenced by maintained photosynthetic function at lower exposure levels and enhanced antioxidant responses. Organic amendments can reduce PAH bioavailability through sorption to organic matter and stimulate microbial degradation processes [22]. Additionally, improved nutrient status may enhance plant stress tolerance capacity. However, the protective effects were insufficient to prevent biochemical perturbations at higher PHE concentrations. This highlights the need for integrated approaches combining organic fertilization with other remediation strategies for severely contaminated soils.
Limitations and Future Research
Several limitations should be acknowledged. The relatively short exposure period (28 days) may not reflect long term impacts of chronic PAH contamination. Additionally, the study examined only one PAH compound, while agricultural soils typically contain complex PAH mixtures with potentially synergistic or antagonistic effects.
Future research should investigate: (1) long-term effects of PAH exposure on crop productivity and quality, (2) accumulation and translocation of PAHs and their metabolites in edible tissues, (3) effectiveness of different organic amendments in mitigating PAH toxicity, and (4) development of integrated soil remediation strategies for contaminated agricultural systems.
Conclusions
This study demonstrates that phenanthrene exposure significantly affects the physiological and biochemical responses of cucumber plants grown in organically fertilized soil. Key findings include:
These findings have important implications for agricultural sustainability and food safety in contaminated environments. While cucumber demonstrated resilience through activated defense mechanisms, the significant biochemical alterations, particularly in fruits, raise concerns about crop quality and consumer safety. The results highlight the need for integrated approaches combining organic soil amendments with other remediation strategies to ensure sustainable crop production in PAH contaminated soils.
Acknowledgments
The authors acknowledge the International Institute of Tropical Agriculture (IITA), Ibadan, for providing cucumber seeds and the technical staff of OAUSTECH for their assistance during the experimental period.
References