Eyuel Kassa1*, Nega Berhane1, Mastewal Birhane2, Mulugeta Ayalew3, Aschalew Gelaw4, Abdulkader Mohamedsaid Gidey5 and Ermias Teklehaimanot6
1Department of Medical Biotechnology, Institute of Biotechnology, University of Gondar, Ethiopia
2University of Gondar, College of Veterinary Medicine and Animal Sciences, Gondar, Ethiopia
3Department of Pediatrics and Child Health University of Gondar, College of Medicine and Health Sciences, School of Medicine, Gondar, Unit of Pediatric Hematology Oncology, Gondar, Ethiopia
4Department of Medical Microbiology, College of Medicine and Health Sciences, School of Biomedical and Laboratory Sciences, University of Gondar, Gondar, Ethiopia
5Department of Pediatrics and Child Health, Addis Ababa University, College of Medicine and Health Sciences, School of Medicine, Addis Ababa, Unit of Pediatric Hematology Oncology, Addis Ababa, Ethiopia
6Department of pathology, University of Gondar, College of Medicine and Health Sciences, School of medicine, Gondar, Ethiopia
*Correspondence: Eyuel Kassa, Department of Medical Biotechnology, Institute of Biotechnology, University of Gondar, Ethiopia, E-mail: eyuel2000@gmail.com
Received: 29 July 2024; Accepted: 20 August 2024; Published: 30 August 2024
Citation: Kassa, Eyuel, et al. “Prevalence of Acute Lymphoblastic Leukemia in Ethiopia: A Systematic Review and Meta-analysis.” J Neonatol Pediat Care (2024): 102.
DOI: 10.59462/JNPC.1.1.102.
Copyright: © 2024 Kassa E. This is an open-ac cess article distributed under the terms of the Cre ative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: There have been very few studies on the prevalence of Acute Lymphocytic Leukemia (ALL) across all age groups in Ethiopia. However, findings on the prevalence of ALL infections vary and are inconsistent. Therefore, this systematic review and meta-analysis aim to provide a pooled prevalence estimate of ALL among pediatric and older adult patients in Ethiopia.
Methods: We systematically searched PubMed, Scopus, Science Direct, Cochrane Library, Google Scholar and Google to retrieve relevant studies. The pooled estimate of ALL was presented using DerSimonian and Laird’s random-effects model with a forest plot. Publication bias was assessed using Egger’s test and a funnel plot. The I-squared statistic was used to test for heterogeneity. Subgroup and sensitivity analyses were also conducted.
Result: This review included seven primary studies with a total sample size of 14,353. Due to significant heterogeneity, a random-effects model was employed. The pooled estimate of overall ALL prevalence was 1% (95% CI: 1.0–2.0). The highest prevalence was 3% in Oromia, while the lowest was 1% reported in Addis Ababa and Amhara National Regional State. Heterogeneity was substantial (I2 = 98.89%; P < 0.000), attributed to geographic location, region and study period. Additionally, ALL prevalence varied significantly between regions and within the country. The meta-analysis revealed considerable inter-study variation and potential publication bias.
Conclusion: The overall prevalence of ALL was found to be low, highlighting the need for improved strategies in developing diagnostic algorithms and treatment protocols for blood cancers. Enhancing awareness, diagnosis and management can help mitigate complications associated with blood cancers. This study is registered with CRD42019147694.
Keywords
Acute lymphocytic leukemia • Children • Adults • Systematic review • Meta-analysis • Ethopia
Introduction
Acute lymphoblastic leukemia (ALL) is characterized by a clonal, aggressive proliferation of lymphoid precursors in the bone marrow, leading to maturation arrest [1]. The resulting clinical picture includes tumor syndrome and bone marrow failure, constituting a medical emergency with a high risk of mortality without prompt intervention. In Europe and North America, acute leukemia is the most prevalent malignant disease in both children and older adults, accounting for a significant portion of childhood cancer deaths [2]. Advances in therapy have increased the cure rate to nearly 80% among children with ALL in these regions [3]. Lymphoblastic acute leukemia, a type of hematological cancer, manifests when white blood cells exhibit abnormal behavior and bone marrow fails to produce adequate platelets and red blood cells. Hematological disorders encompass a range of conditions, both malignant and non-malignant [4]. In individuals under 20 years old, leukemia is the leading cause of cancer-related mortality. Among adults, leukemia accounts for 1.2% of all cancer cases globally. Between 2007 and 2017, the incidence of leukemia worldwide rose by 19%, with a 13% increase in leukemia-related fatalities [5].
Leukemia encompasses several types categorized broadly into acute (rapidly worsening) and chronic (slowly worsening), affecting lymphoid and myeloid white blood cell lineages. The French-American-British (FAB) and World Health Organization (WHO) classifications categorize leukemia based on morphology, cytochemistry, immunophenotyping, cytogenetics and clinical features, highlighting the diverse nature of the disease. Although any form of leukemia can occur in children, ALL is the most prevalent [6]. The etiology of leukemia is multifactorial, involving genetic and environmental influences. Acquired DNA abnormalities in leukemia cells lead to uncontrolled growth and loss of normal white blood cell functions. Chromosomal translocations, such as the BCR-ABL1 fusion in chronic myeloid leukemia (CML) and some cases of ALL, contribute to oncogene formation [7]. Genetic predispositions, including Down syndrome and other inherited syndromes, as well as exposures to ionizing radiation, chemicals, therapeutic drugs and viral agents, are implicated in leukemia development [4,8]. Leukemia ranks among the 8th to 12th most common cancers globally. In Ethiopia, leukemia, particularly ALL, is prevalent among children, constituting 89% of pediatric leukemia cases Challenges such as limited medical resources and high mortality rates highlight the urgent need for improved healthcare infrastructure and access to treatment [9].
Data on children ALL are extremely rare in sub-Saharan Africa due to a lack of trained medical personnel and a lack of adequate diagnostic facilities. In 2013, after lymphoma, retinoblastoma and nephroblastoma, ALL was the fourth most common pediatric cancer, according to a previous study. With a survival rate that barely ever reaches 15%, the prognosis is still bleak in the majority of African countries. This high mortality is typically caused by a delay in care and a high drop-out rate [1,10]. To address these gaps, meta-analysis serves as a powerful tool for synthesizing diverse datasets from multiple studies, thereby enhancing reliability, generalizability and statistical power of the findings. The wealth of data on ALL has not only yielded more robust and credible information but also facilitated new biological insights. The limited adoption of national cancer registries may stem from governmental non-mandates on reporting, leading some institutions to establish their own registries, thereby compromising data consistency. This study aims to conduct a comprehensive review and meta-analysis on the prevalence of childhood ALL and ALL among older adults in Ethiopia, aiming to provide valuable insights into the disease’s landscape.
Methods
Objectives
The primary objectives of this review were to assess the pooled prevalence rates of childhood ALL and older adults and to investigate potential regional variations within Ethiopia.
Reporting and registration of the protocol
The reporting adhered to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines [11]. The protocol was registered with the International Prospective Register of Systematic Reviews (PROSPERO) under protocol number CRD43019107674, accessible at https://www??/PROSPERO.
Study design
Systematic review and meta-analysis were conducted following the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines [12]. The PRISMA checklist was utilized to ensure comprehensive reporting of relevant information in the analysis.
Eligibility criteria
To establish criteria for inclusion and exclusion in prevalence studies, the CoCoPop (Condition, Context and Population) technique was employed.
Inclusion criteria
1.Setting: Studies conducted within Ethiopia, including its administrative regional states and city administrations.
2.Population: Pediatric children and older adults diagnosed with ALL.
3.Study Type: Cross-sectional studies reporting on the prevalence of ALL.
4. Outcome: Prevalence of acute lymphoblastic leukemia among pediatric and adult populations.
5.Publication Status: All published studies.
6.Publication Year: No restriction on publication year. 7.Publication Date: Studies published up to January 1982. 8. Language: Studies published in English only. Exclusion criteria
Articles that could not be accessed despite two email attempts to contact the principal investigator, as well as citations without abstracts (such as case reports, case series, editorials and qualitative studies reporting sex specific findings), were excluded from the final analysis.
Data sources
Primary studies relevant to the systematic review and meta-analysis of ALL across all age groups were identified through comprehensive searches of electronic databases including PubMed, Scopus, Science Direct, Cochrane Library, Google Scholar and direct Google searches.
Searching strategies
Our search strategy included a combination of terms and phrases related to ALL among pediatric patients and adults affected in Ethiopia. Key search terms and phrases used were: prevalence, proportion, incidence, magnitude, epidemiology, blood cancer, hematologic malignancy, leukemia, acute leukemia, childhood leukemia, lymphocytic leukemia, magnitude of acute lymphoblastic leukemia. Corresponding authors were contacted to obtain full-texts for studies with incomplete data. Additionally, relevant studies were identified by reviewing reference lists of included studies to ensure comprehensive coverage beyond electronic database searches. Advanced search techniques for different electronic databases utilized a combination of MeSH (Medical Subject Heading) terms and Boolean operators (AND, OR). For PubMed, the search strategy was structured as follows: (prevalence [Title/Abstract] OR proportion [Title/Abstract] OR incidence [Title/Abstract] OR epidemiology [Title/Abstract] OR blood cancer [Title/Abstract] OR hematologic malignancy [Title/ Abstract] OR leukemia [Title/Abstract] OR acute leukemia [Title/Abstract] OR acute lymphoblastic leukemia [Title/ Abstract] OR childhood leukemia [Title/Abstract] OR lymphocytic leukemia [Title/Abstract]) AND (in Ethiopia [Title/Abstract]).
Definition of reviewed variables
This study reviewed original data on the incidence/ prevalence of acute lymphoblastic leukemia in general, separately in men and women, obtained from studies conducted in Ethiopia without time limitation.
Selection of studies
All retrieved primary studies were exported to Endnote version 7 for deduplication and management of citations and bibliography. Two independent reviewers (EK and MB) screened the studies based on specific selection criteria. Discrepancies were resolved through consensus between the two reviewers (EK and MB) after thorough discussion. In cases of persistent disagreement, a third reviewer (AG, MA and ET) arbitrated based on established article selection criteria.
Risk of bias assessment
Each included study underwent independent assessment by two authors (EK and MB) using a quality assessment tool adapted from Hoy et al. [13] to evaluate both external and internal validity. The tool consists of 9 items for assessing bias, with scores categorized as low risk (0–3), moderate risk (4–6) and high risk [7-9].
Data abstraction
Data extraction was independently conducted by two authors (EK & MB) using a structured data extraction format in Microsoft Excel 2010. After independent extraction, discrepancies were reconciled by the two authors (EK, MA and MB), with input from a third reviewer (AG) as necessary. Corresponding authors of eligible studies were contacted via email and/or phone to obtain missing data. Extracted data included: corresponding author’s name, publication year, publication date, study design (cross sectional), study location, sample size, study type, assay method, overall prevalence of ALL and individual incidence or proportion of ALL.
Outcome measurement
This review considered studies reporting the prevalence of ALL among pediatric and adult populations in Ethiopia as the primary outcome. Prevalence was defined as the presence of blast cells in bone marrow smear or biopsy and peripheral morphology of pediatric and adult patients across all age groups of ALL patients, using Wright stain/H & E staining or Sudan Black B (SBB) staining method to define prevalence [14].
Quality assessment
The quality of prevalence studies was assessed using adapted Joanna Briggs Institute (JBI) criteria [15]. Criteria included: appropriate sample frame, sampling technique, sample size adequacy, description of study subjects and setting, coverage of data analysis, validity of condition identification method, reliability of measurement, appropriateness of statistical analysis and adequate response rate management.
Reliability
The second reviewer (MB) was blinded to primary reviewer (EK) decisions during study selection, data extraction and bias assessment. Discrepancies were resolved through discussion between reviewers, with arbitration by a third reviewer (AG) if needed.
Statistical analysis
Statistical analysis was conducted by three authors (EK, MB and Prof. NB). Funnel plots and Egger’s test were used to assess publication bias likelihood. Heterogeneity was assessed using I-squared statistics, with values of 25%, 50% and 75% indicating low, moderate and high heterogeneity respectively (significant heterogeneity defined as I2 > 75%). The pooled prevalence of ALL was estimated using DerSimonian and Laird’s random-effects model [16,17] Subgroup analysis and sensitivity analysis were performed to explore potential outlier studies and assess their impact on overall estimates. Stata version 14 and installed meta-analysis packages were utilized for analysis, presenting outcomes with point estimates and 95% confidence intervals
Results
The initial database search yielded 532 articles. After removing duplicates based on title and abstract, 34 articles remained for full-text examination. Following the exclusion of 27 articles during this phase, seven publications were included in both the systematic review and meta-analysis (Figure 1). Four of these studies were conducted in the Amhara National Regional State, two in Addis Ababa and one in Oromia.
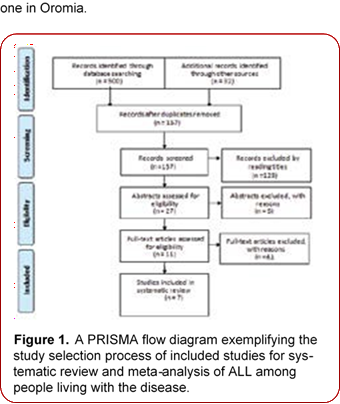
Figure 1. A PRISMA flow diagram exemplifying the study selection process of included studies for sys tematic review and meta-analysis of ALL among people living with the disease.
Characteristics of included studies
After thoroughly searching major electronic databases, we retrieved 532 studies. Ultimately, seven studies [4,9,10,18,19,20,21] with a total sample size of 14,353 pediatric children and adults living with ALL infections were included for qualitative synthesis and meta-analysis (Table 1). The sample sizes of the included studies ranged from 82 [9] to 7969 [19]. We intended to include all types of observational studies (case-control, case series, cohort and cross-sectional studies), but only cross-sectional studies that met the inclusion criteria were found and included in the analysis. Quality assessment was conducted using the Hoy et al. critical appraisal tool, revealing a considerable risk of bias across the studies. All studies included participants across all age groups and some studies reported on the sex distribution of participants.
The pooled estimate of ALL prevalence among the included studies was 1% (95% CI: 1–2%). Significant heterogeneity was observed across the studies (I2 = 98.89%). To address this variability, a random-effects model was used for the meta-analysis, yielding an estimated pooled prevalence of overall acute lymphoblastic leukemia at 1% (95% CI: 1.00–2.00) (Figure 2). The seven studies included in this meta-analysis and comprehensive review (Table 1) was conducted in various regions of Ethiopia, including Amhara(University of Gondar Comprehensive Specialized Hospital, Bahrdar Tibebe Gion Hospital, Wolo University Teaching Hospital), Oromia (Jima University Teaching Hospital) and Addis Ababa (Black Lion Specialized Hospital). These studies were published between 1994 and 2022. The prevalence of ALL varied significantly among regional states, with the highest prevalence observed in Oromia (Jima) at 3% and the lowest prevalence reported in Addis Ababa and Amhara National Regional State at 1%. Each study utilized a cross-sectional design and bone marrow was the primary method for ALL diagnosis, employing various staining techniques such as Sudan Black B, Wright’s or Wright-Giemsa stains and Hematoxylin and Eosin staining methods.
Methodological quality of studies
According to JBI standards for assessing the quality of primary research, it is recommended to include primary studies in the meta-analysis if they received a score of 58% or higher (Table 2). Two studies scored above 70% (Jima and Dessie). The ratings for the remaining trials ranged from 60% to 70%. Sample sizes in the vast majority of studies (79%) were deemed appropriate, ranging from 82 to 7969 participants [4,9,10,18,19,20,21]. Importantly, all included trials utilized bone marrow, a reliable technique for diagnosing ALL infections.
|
Author |
Publication year |
Study- period |
Design |
Country |
Region |
Age |
Sample size |
Event |
Setting |
Type |
Assay method |
|
-18 |
2021 |
2019 |
CS |
Ethiopia |
Amhara |
0-18 |
1257 |
2 |
Mixed |
ALL |
BM |
|
(10). |
2020 |
2019 |
CS |
Ethiopia |
Oromia |
0-50 |
332 |
9 |
Mixed |
ALL |
BM |
|
-19 |
1994 |
1982- 1992 |
CS |
Ethiopia |
AA |
13–78 |
82 |
37 |
Mixed |
ALL |
BM |
|
-9 |
1990 |
1982- 1987 |
CS |
Ethiopia |
AA |
14-80 |
7969 |
95 |
mixed |
ALL |
BM |
|
(20). |
2021 |
2015 - 2019 |
retrospective |
Ethiopia |
Amhara |
1–90 |
1342 |
22 |
Mixed |
ALL |
BM |
|
-4 |
2022 |
2020- 2021 |
CS |
Ethiopia |
Amhara |
Jan- 75 |
228 |
2 |
Mixed |
ALL |
BM |
|
-21 |
2016 |
2011- 2014 |
CS |
Ethiopia |
Amhara |
0-14 |
4400 |
33 |
mixed |
ALL |
BM |
Table 1. A summary of a descriptive characteristics and quality status of the included studies for this systematic review and meta-analysis.
|
Study |
Q1 |
Q2 |
Q3 |
Q4 |
Q5 |
Q6 |
Q7 |
Q8 |
|
|
Total |
|
|
Q9 |
Y |
N |
U |
|||||||||
|
-18 |
Y |
N |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
8 |
1 |
0 |
|
(10). |
Y |
N |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
8 |
1 |
0 |
|
-19 |
Y |
N |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
8 |
1 |
0 |
|
-9 |
Y |
N |
N |
Y |
Y |
Y |
Y |
Y |
U |
6 |
3 |
1 |
|
(20). |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
N |
8 |
1 |
0 |
|
-4 |
Y |
Y |
Y |
N |
Y |
Y |
Y |
Y |
Y |
8 |
1 |
0 |
|
-21 |
Y |
N |
Y |
Y |
Y |
Y |
Y |
Y |
Y |
7 |
1 |
0 |
Table 2.
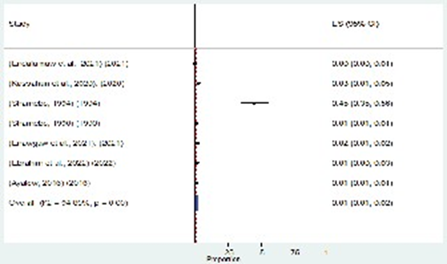
Figure 2. Forest plot for the prevalence of ALL diseases in Ethiopia.
Handling heterogeneity
The random effects model’s pooled estimate exhibits significant heterogeneity. To address this heterogeneity, sensitivity analysis and subgroup analysis were conducted. A number of heterogeneity-related factors, such as publication year, sample size and geography, were examined using subgroup analysis. The assay method, study design and setting for the variables, however, was statistically significant.
Subgroup analysis
Despite conducting subgroup analysis across the nation’s administrative areas, factors contributing to heterogeneity such as age categories and place of residence or study area were considered. The subgroup analysis indicated that
Oromia had the highest prevalence of ALL, reported at 3% (95% CI: 1.00-5.00), followed by Addis Abeba and Amhara, each with 1% prevalence (95% CI: 1.00-1.00). The study stratified infections across all patient age groups based on various locations in Ethiopia, revealing significant variation (I2 = 94.89, P = 0.00) (Figure 3). Moreover, the prevalence estimate for Oromia region is 3.0%, falling within the range of 1.00 to 5.00. There is notable regional disparity: Amhara and Addis Abeba exhibit lower prevalence compared to Oromia, which shows the highest prevalence.
Sensitivity analysis
We conducted a sensitivity analysis by excluding a study with a limited sample size. The analysis revealed substantial heterogeneity, with an I2 of 98.89% and a P value of 0.000. The overall pooled prevalence remained
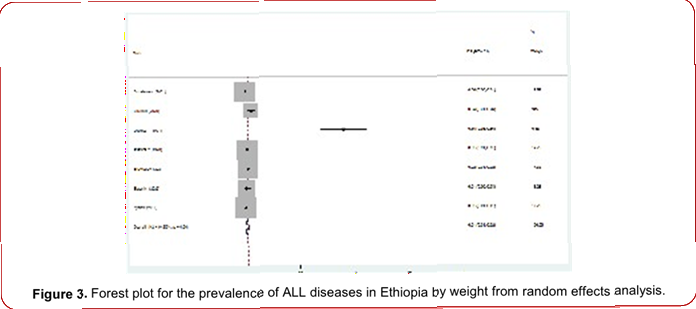
Figure 3. Forest plot for the prevalence of ALL diseases in Ethiopia by weight from random effects analysis.at 1.0% (95% confidence interval [1.0-2.0]). To assess the impact of individual studies on the outcome, we employed the leave-one-out method. Sequentially excluding each study did not alter the overall prevalence significantly, indicating robustness in our findings (Figure 4).
Publication bias
Using a funnel plot and Egger’s test, we evaluated the publication bias of the studies. An element of publication bias existed in this systematic review. When all studies were considered, there was evidence of an Egger’s test p-value of 0.000 (Egger’s test showed that publication bias was statistically significant for studies reporting the prevalence of ALL cancers; p = 0.00). (Figure 5,6)
The estimated pooled prevalence of individual ALL patients
Seven studies utilized the fixed-effect model to present their estimated pooled prevalence of ALL, revealing significant inter-study heterogeneity. Consequently, a random-effects model was employed to determine the pooled prevalence. The distribution of effect sizes was normalized using a double arcsine transformation. The evaluation noted a significant increase in the prevalence of ALL between studies conducted from 1982 to 1992 and 2019, with a subsequent decrease observed in a 2022 study. In the random-effects model, the prevalence of ALL was estimated at 1% (95% CI: 1-2%), reflecting significant inter-study variability.
Discussion
This study represents the first systematic review and meta-analysis on the prevalence of ALL in Ethiopia. Seven studies were reviewed and analyzed to estimate the pooled prevalence of ALL. Cancer, as a non-communicable disease, contributes significantly to global morbidity, mortality and economic burden [22]. The prevalence of ALL is increasing in developing countries, contrasting with trends in developed nations [23]. In Ethiopia, cancer has become the second leading cause of death among adults (24). Factors such as exposure to certain chemicals, family history, age and radiation exposure contribute to this burden (25). Despite an increasing burden, childhood and older adult ALL cancers were poorly quantified and continued to receive low public health priority in Ethiopia, largely because of the overwhelming burden of communicable diseases and limited resources. Hence, we aimed to determine the prevalence of childhood ALL cancer, older adults and predictors among mother-child pairs in four referral hospitals in Ethiopia. In this study, the overall pooled prevalence of ALL was 1% (95% CI: 1-2%).
This finding is in line with 1.7% in USA [26]. The other finding is a bit lower than studies conducted in Thiland between ages 11 and 40 years, that ranged between 25.8% to 26.2% [27]and 4.32% in Indonesia to 4.32% in Nordic [28] and 7.3% to 57.8% in Indian [29]. The discrepancy might be due to differences in the diagnostic capacity of
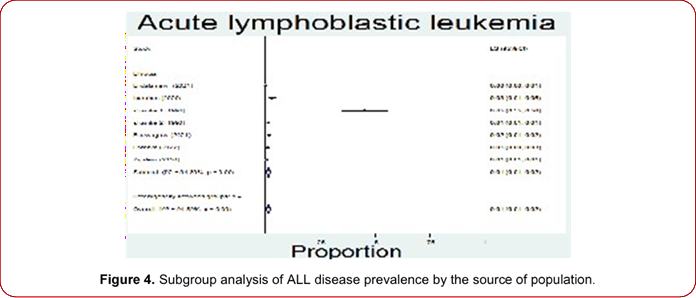
Figure 4. Subgroup analysis of ALL disease prevalence by the source of population.
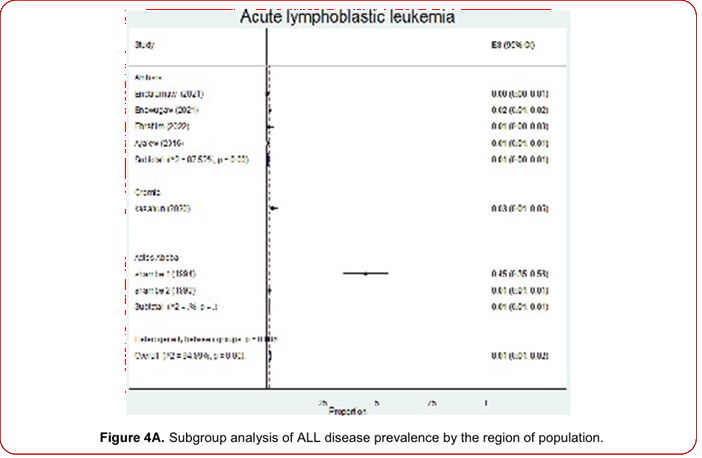
Figure 4A. Subgroup analysis of ALL disease prevalence by the region of population.
the health institutions or availability of diagnostic facilities, health-seeking behavior or norms of the population and perhaps access to health services; the other reasons might include misdiagnosis, underreporting, or pre-emptive death from infectious disease [25]. For instance, only 23% of health facilities offer services for cancer in Ethiopia [30].
Accurate epidemiological data on hematologic or pediatric cancers will enable the mobilization of sufficient resources for proper screening, prevention and treatment of cancers. Most of the ALL patients with blood cancer in our analysis resided in the urban area of the study setting due to the proximity and accessibility of medical resources. But a
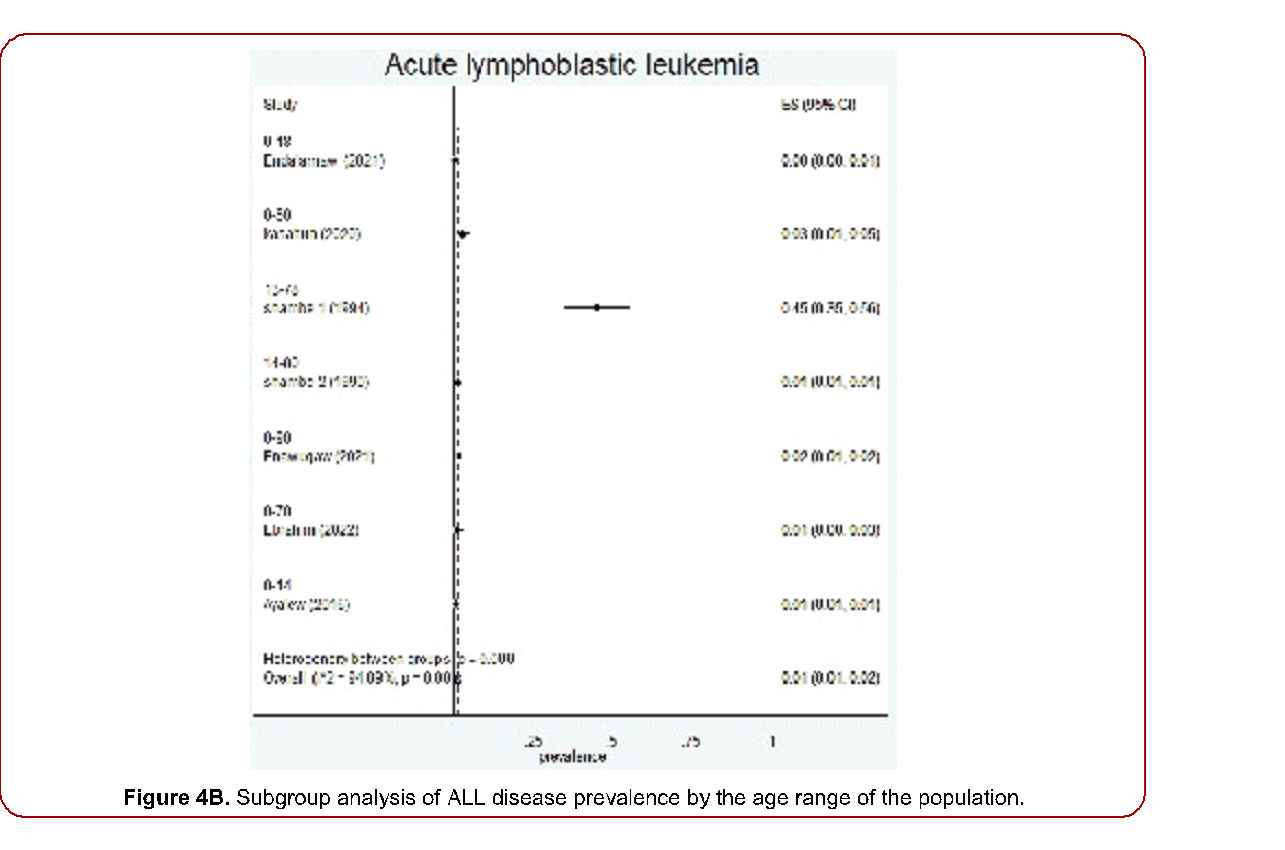
Figure 4B. Subgroup analysis of ALL disease prevalence by the age range of the population.
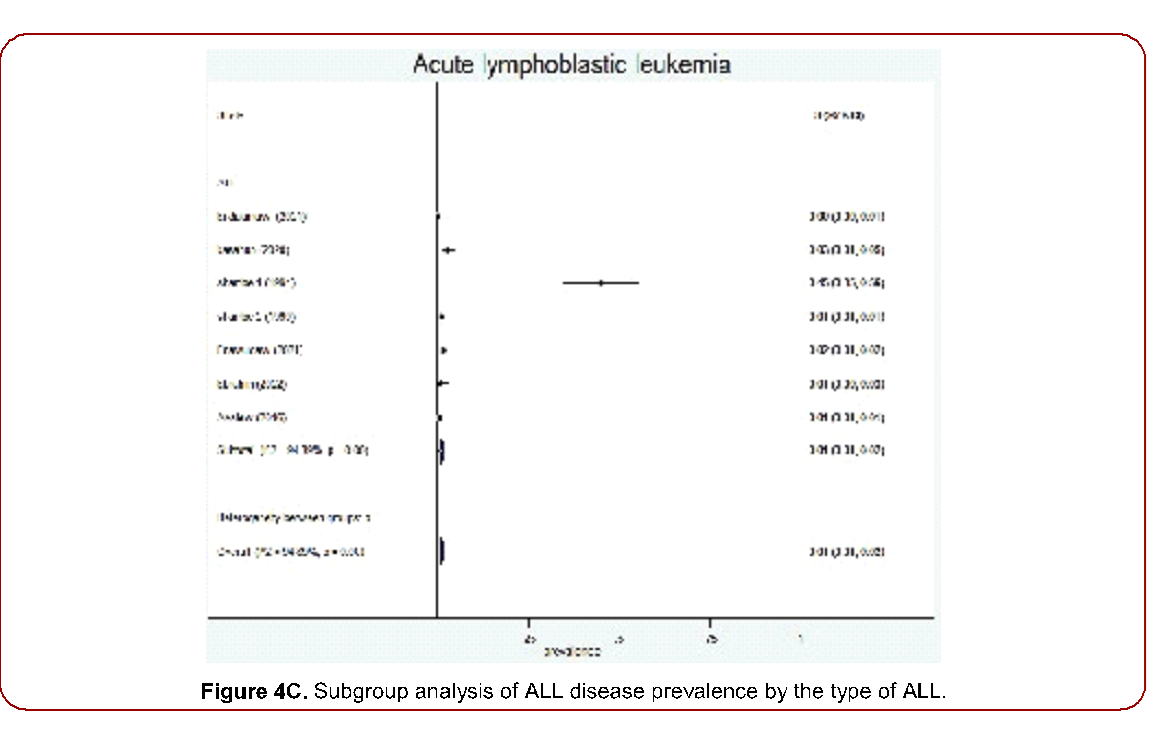
Figure 4C. Subgroup analysis of ALL disease prevalence by the type of ALL.
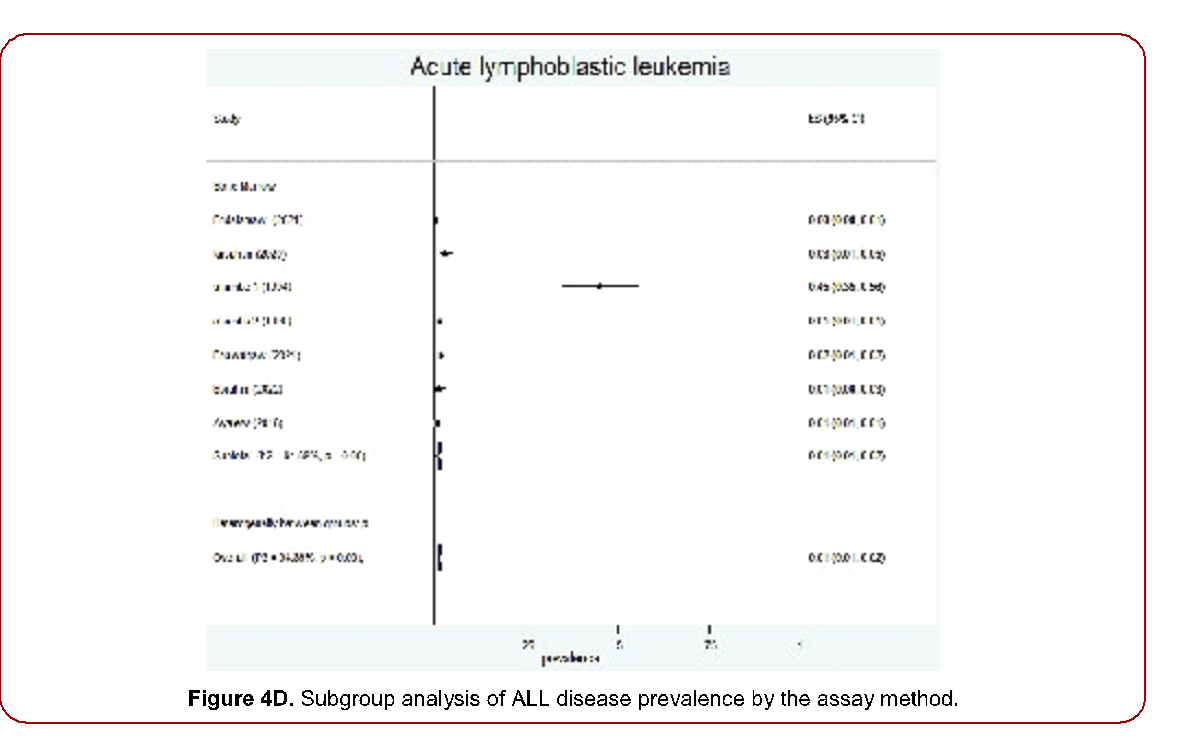
Figure 4D. Subgroup analysis of ALL disease prevalence by the assay method.
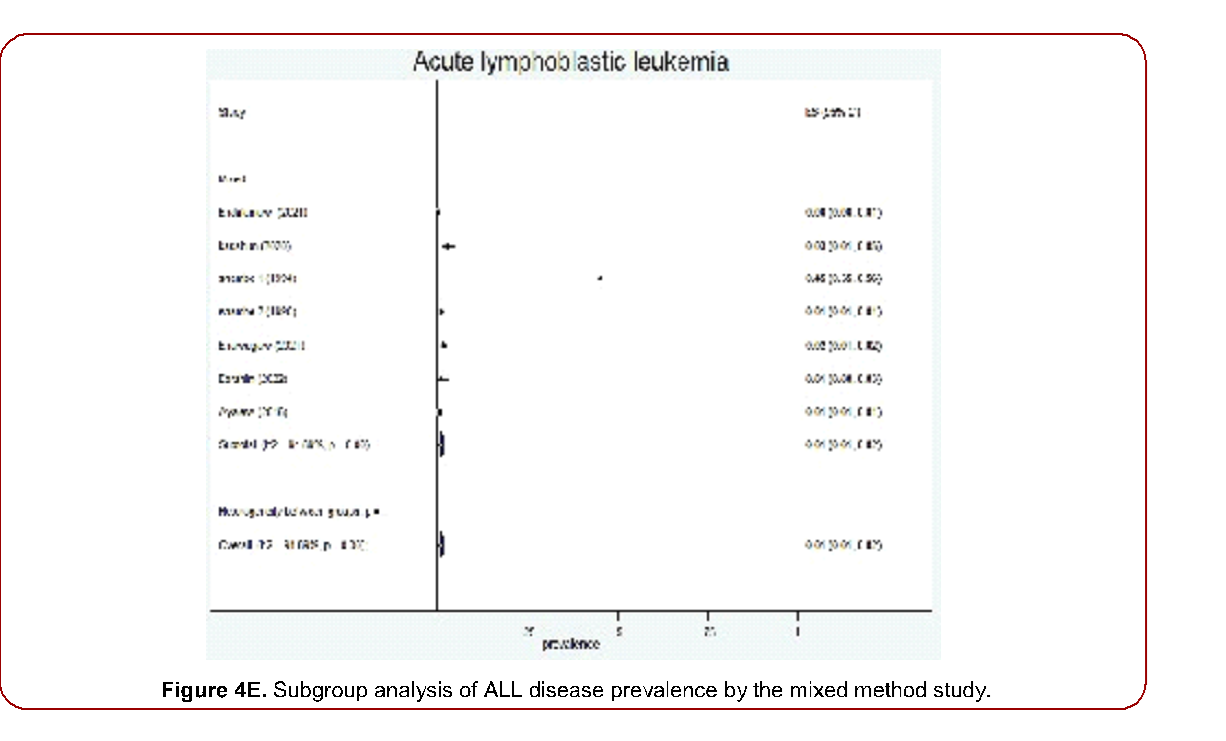
Figure 4E. Subgroup analysis of ALL disease prevalence by the mixed method study.
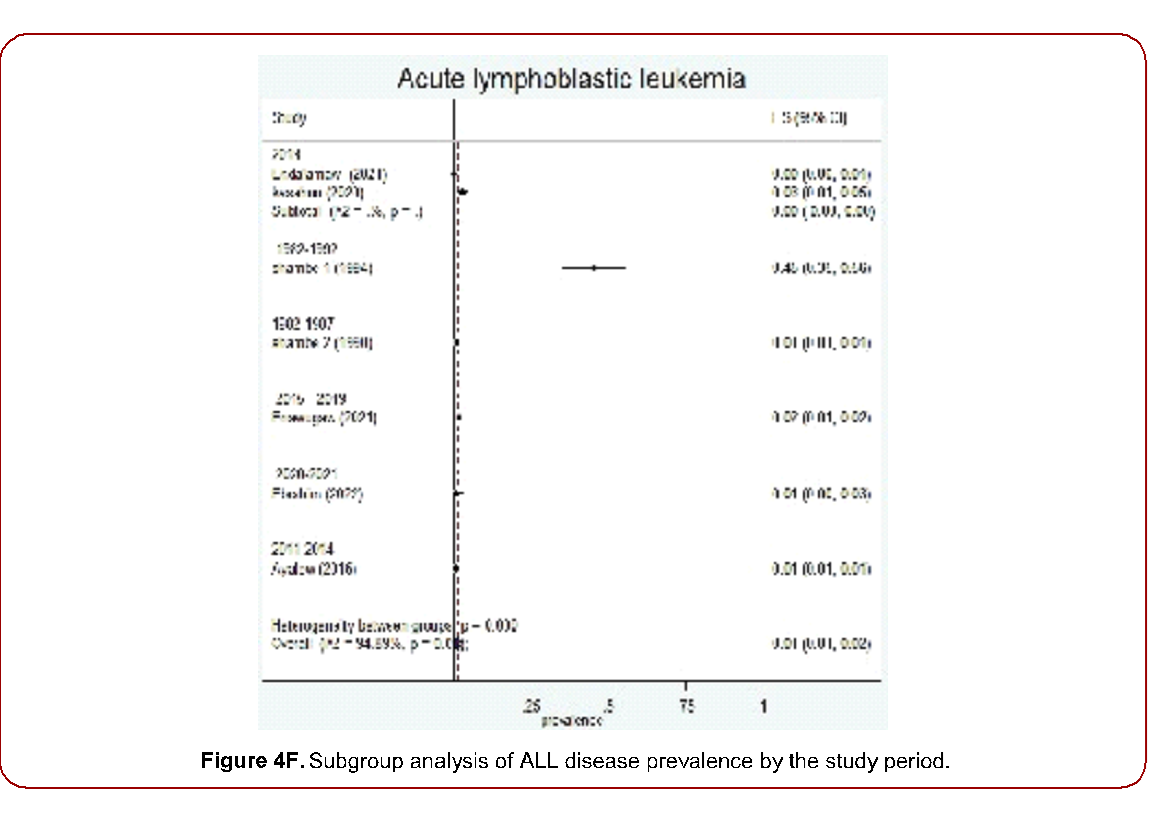
Figure 4F. Subgroup analysis of ALL disease prevalence by the study period.
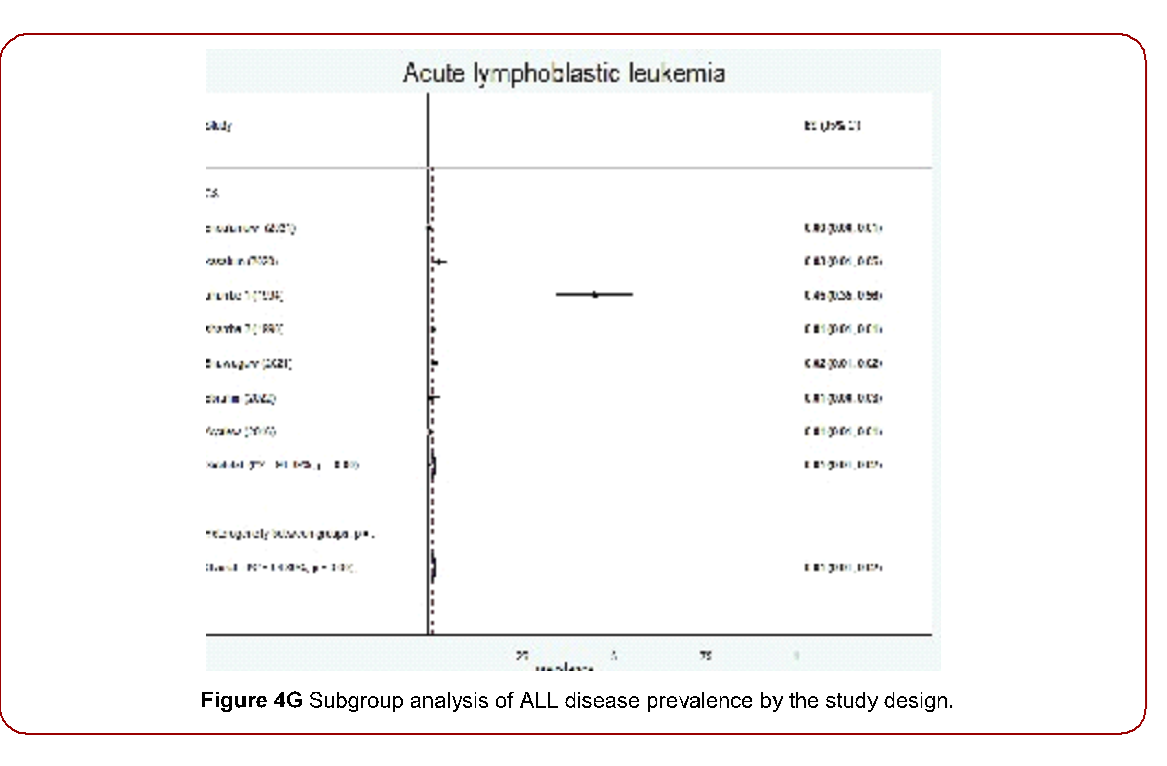
Figure 4G Subgroup analysis of ALL disease prevalence by the study design.
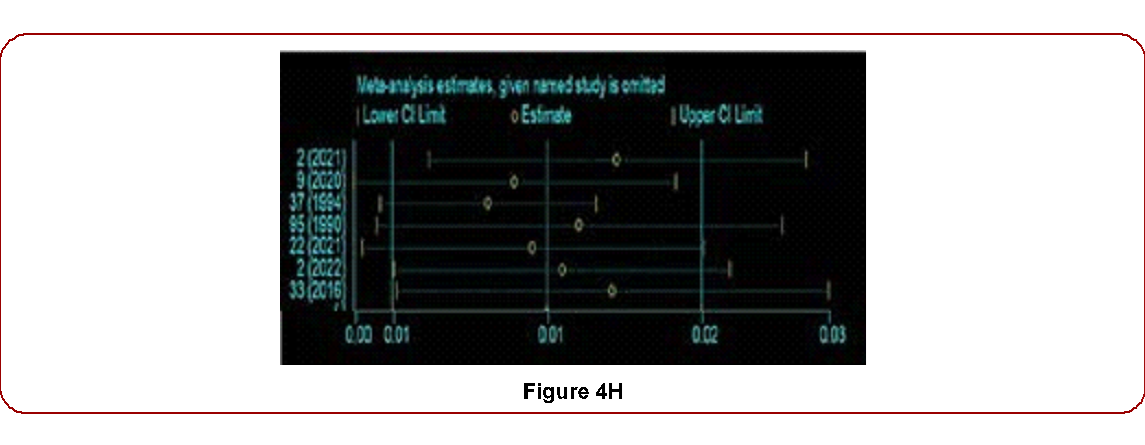
Figure 4H
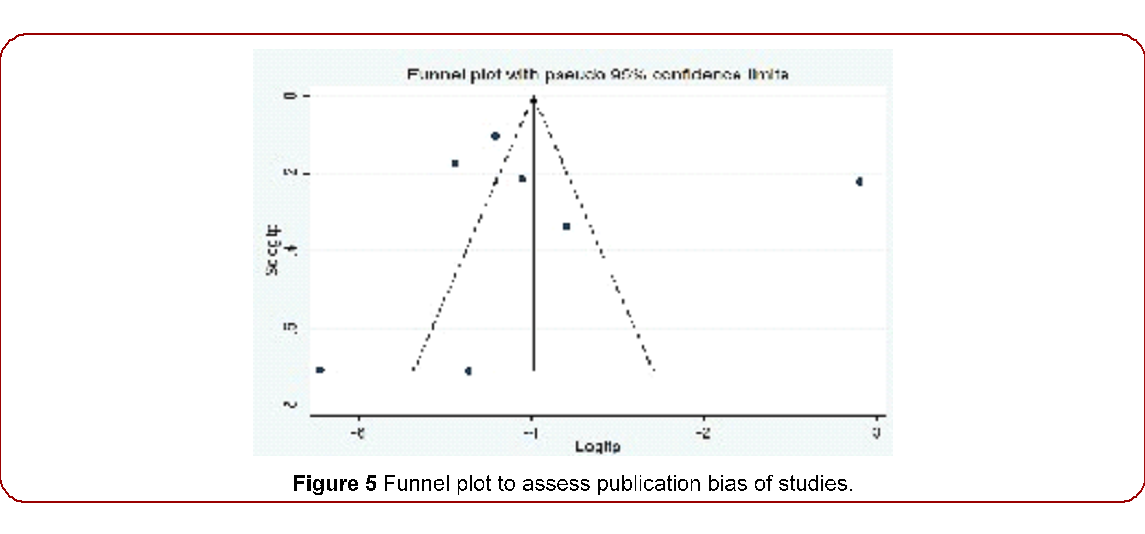
Figure 5 Funnel plot to assess publication bias of studies.
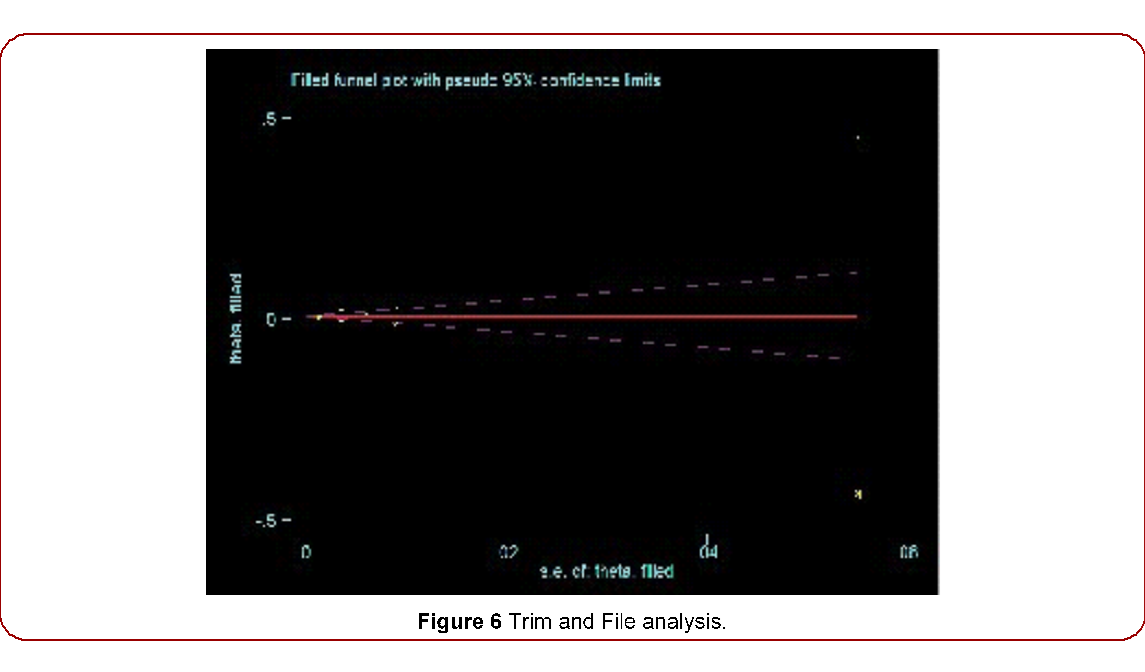
Figure 6 Trim and File analysis.
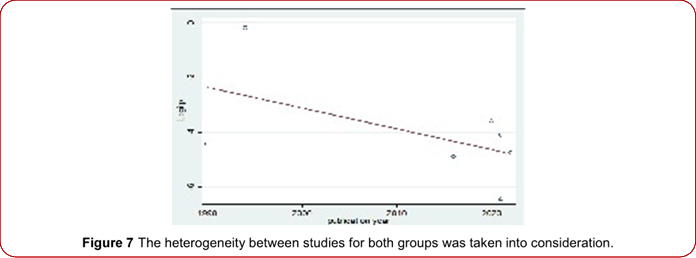
Figure 7 The heterogeneity between studies for both groups was taken into consideration.cancer patient from a rural area might have died before registration or diagnosis as access to health care is very deficient. Hence, improved diagnostics and suspicion on clinical examination might result in an increasing number of patients diagnosed with cancer in rural areas. The WHO global initiative for childhood and older adult cancer aims to increase the overall survival for six key cancers (acute lymphoblastic leukemia, Burkitt’s lymphoma, Burkitt’s lymphoma, Hodgkin’s lymphoma, low-grade glioma, retinoblastoma and Wilms tumor) to 60% globally by 2030 through the development of national centers of excellence and expanding regional satellites [31]. Furthermore, child survival is highly dependent on several health indicators, including the number of physicians and nurses per 1000 population, infrastructure, human resources and level of supportive care [32]. However, from more than 446 hospitals in Ethiopia, only less than five centers were found to have a pediatric-specific cancer center, which makes it very difficult to access early diagnosis and treatment. This will have ended with poor survival for children and older adults with cancer.
Our systematic analysis of ALL literature in Ethiopia revealed an inconsistent trend in prevalence. There was an increase in ALL prevalence from 1990 to 2000, followed by a decline from 2010 to 2022 (Figure 7). However, this difference in trend was not statistically significant. Globally, the prevalence and mortality of ALL have been observed to increase in developing countries [33,34]. Consistently, studies from sub-Saharan Africa, including Ethiopia, indicate that ALL prevalence and associated mortality have not decreased [24]. Based on geographical distribution,the highest prevalence of ALL was observed in the Oromia region, followed by the Addis Ababa and Amhara National Regional States. However, attributing the highest pooled prevalence to Oromia was challenging due to significant heterogeneity. There was variation in ALL prevalence in Amhara and Addis Ababa, likely due to differences in population demographics. Specifically, [21] focused solely on the pediatric population (median age 4.2 years), while [20] examined both pediatric and adult populations
Significant heterogeneity was detected across studies in these prevalence estimates, prompting cautious interpretation of the results. The heterogeneity remained substantial (I2 = 98.89%). The primary limitation of this systematic review and meta-analysis was the high between-study heterogeneity. Many analyses, including the main analysis of pooled ALL prevalence across the seven studies, showed I2 > 75%. Methodological differences in ALL detection and variations in background populations (especially across different age groups) were likely major contributors to this study-to-study variation. Additionally, the precision of several subgroup and outcome analyses was restricted by the relatively small number of included studies. Children and older adults with blood cancer in Sub-Saharan Africa face significant challenges. Children in low-income countries often present with advanced-stage disease due to limited healthcare facilities, transportation issues, lack of disease information and treatment options [36]. Similarly, in Ethiopia, previous studies have highlighted prolonged hospitalizations for cancer patients due to shortages in anticancer drugs, as well as inadequate diagnostic and treatment facilities.Despite Ethiopia having nine regions, the majority of studies in this review focused on Oromia, Amhara and Addis Ababa. Moreover, the systematic review and meta analysis included a limited number of studies. Considering that these regions account for 80% of the population [37] caution is necessary when generalizing the findings to the Ethiopian population.
Quality of evidence
According to the GRADE criteria, the overall quality of evidence is likely low to very low. Initial quality of all observational studies is low. Given the substantial variation in point prevalence among studies, the narrow confidence intervals around pooled prevalence estimates and significant heterogeneity observed, this assessment appears accurate. It suggests that the effect estimate is provisional and subject to change with further studies. The following suggestions are provided to enhance the quality of future research.
Strengths
In the context of this comprehensive review, to our knowledge, this is the first systematic review and meta analysis of national ALL prevalence among pediatric and older adult populations during the study period. Consequently, the study elucidates the epidemiology of ALL in diverse settings. Strength of this study is its potential as a foundational resource for future investigations and as informative material for healthcare professionals and stakeholders in this field. Our search yielded an unexpectedly high number of studies, allowing us to rigorously select studies of high quality for full analysis. Although high-risk bias studies were excluded from primary analyses, a secondary comparison was conducted between high-risk and medium- or low-risk bias studies.
Study limitations
One limitation is that the study was institution-based, focusing on ALL patients visiting outpatient departments for various illnesses, which may not represent children who appear healthy but may have undiagnosed blood cancers in the community. This limitation stemmed from feasibility constraints in terms of resources. The study results should be interpreted considering these potential limitations. Additionally, due to limited studies in Ethiopia, generalizing findings to the entire population might be challenging. Inter-study heterogeneity was significant in this review, unresolved by subgroup analysis and influential factors for outcomes could not be definitively identified. In Ethiopia, leukemia research is generally underexplored and all seven studies included in this review were mixed studies involving other hematologic cancers.
Conclusion
Despite childhood and older adult blood cancers receiving limited attention from local policymakers, their prevalence remains significant in the study setting. Increased awareness, knowledge, diagnostic tools and affordability may lead to higher reported cases in the future. International collaborations have played a crucial role in improving the diagnosis and treatment of ALL in Ethiopia. There is a strong recommendation for more efficient implementation of blood cancer diagnostic laboratories in numerous healthcare centers across the country. Notably, Ethiopia faces challenges with its sparse cancer registry data, highlighting the urgent need for better development of regional and national registries. Therefore, this systematic review and meta-analysis offer valuable insights for policymakers, health professionals and researchers to address these gaps. A large-scale community-based study should be undertaken to reach cases that have not accessed treatment.
Declaration
Data Availability
All data generated or analyzed during this study are included within the manuscript. The data supporting the findings of this study are not openly available due to reasons of sensitivity. However, the datasets used and/or analyzed (SPSS data) are available from the corresponding author upon reasonable request.
Funding
This study received no specific grant from any funding agency.
Acknowledgment
The authors are thankful to the University of Gondar Comprehensive Specialized Hospital, for their kind cooperation in facilitating this study. The authors are also grateful to the data collectors for their participation and contribution.
Abbreviations
ALL Acute Lymphoblastic Leukemia CI Confidence Interval
Ph Philadelphia
FAB The French-American-British WHO World Health Organization
PRISMA Preferred Reporting Items for Systematic Review and Meta-Analysis
Guideline
PROSPERO International Prospective Register of Systematic Reviews
CoCoPop Condition, Context and Population MeSH Medical Subject Heading
H&E stain Hematoxylin and Eosin Stain SBB Sudan Balk B
JBI Adapted Joanna Briggs Institute DNA Deoxyribonucleic Acid
WBC White Blood Cells
AML Acute Myelogenous Leukemia CML Chronic Myelogenous Leukemia.
References
1. Togo, B., T. Fousseyni, A. K. Doumbia, and P. Togo. “Childhood acute lymphoblastic leukemia in sub Saharan Africa: 4 years? Experience at the pediatric oncology unit Bamako, Mali.” J Child Adolesc Health 2 (2018): 24-6.
2. Glass, Deborah C., Alison Reid, Helen D. Bailey and Elizabeth Milne, et al. “Risk of childhood acute lymphoblastic leukaemia following parental occupational exposure to pesticides.” J Occup Med Title 69 (2012): 846-849.
3. Domenech, Carine, Xavier Thomas, Sylvie Chabaud and Andre Baruchel, et al. “l‐asparaginase loaded red blood cells in refractory or relapsing acute lymphoblastic leukaemia in children and adults: results of the GRASPALL 2005‐01 randomized trial.” Br J Haematol 153 (2011): 58-65.
4. Ebrahim, Hussen, Temesgen Fisha, Habtu Debash,and Habtye Bisetegn. “Patterns of bone marrow confirmed malignant and non-malignant hematological disorders in patients with abnormal hematological parameters in Northeast Ethiopia.” J Blood Med (2022): 51-60.
5. Boonhat, Hathaichon, and Ro-Ting Lin. “Association between leukemia incidence and mortality and residential petrochemical exposure: A systematic review and meta-analysis.” Environ Int 145 (2020): 106090.
6. H. Tran, Yvette, Scott L. Coven, Seho Park and Eneida A. Mendonca. “Social determinants of health and pediatric cancer survival: a systematic review.” Pediatr Blood Cancer 69 (2022): e29546.
7. Pui, Ching-Hon, Kathryn G. Roberts, Jun J. Yang, and Charles G. Mullighan. “Philadelphia chromosome– like acute lymphoblastic leukemia.” Clin Lymphoma Myeloma Leuk 17 (2017): 464-470.
8. Bassan, Renato, Monika Brüggemann, Hoi-Shen Radcliffe and Elizabeth Hartfield, et al. “A systematic literature review and meta-analysis of minimal residual disease as a prognostic indicator in adult B-cell acute lymphoblastic leukemia.” Haematologica 104 (2019): 2028.
9. Shamebo, Milkias. “Leukaemia in adult Ethiopians.” Ethiop Med J 28 (1990): 31-37.
10. Kassahun, Woldeteklehaymanot, Girum Tesfaye, Lealem Gedefaw Bimerew and Diriba Fufa, et al. “Prevalence of Leukemia and Associated Factors among Patients with Abnormal Hematological Parameters in Jimma Medical Center, Southwest Ethiopia: A Cross‐Sectional Study.” Adv in Hematol 2020 (2020): 2014152.
11. Christophers, Briana, Benjamin Gallo Marin, Rocío Oliva and Weston T. Powell, et al. “Trends in clinical presentation of children with COVID-19: A systematic review of individual participant data.” Pediatric res 91 (2022): 494-501.
12. Bakshi, Pooja R., and Vaishali Y. Londhe. “Widespread applications of host-guest interactive cyclodextrin functionalized polymer nanocomposites: Its meta analysis and review.” Carbohydr Polym 242 (2020): 116430.
13. Van Timmeren, E. A., C. P. Van der Schans, A. A. J. Van der Putten, W. P. Krijnen and H. A. Steenbergen, et al. “Physical health issues in adults with severe or profound intellectual and motor disabilities: A systematic review of cross‐sectional studies.” J Intellect Disabil Res 61 (2017): 30-49.
14. Sushma, A., Venkata Suseela Ch, B. Hepsi, Venkata Reddy, and P. Venkatesh. “Measures of disease frequency and prevalence of drug therapy related problems on basis of prospective observational study in rural hospitalization patients in Nellore.” Int J Healthc (2022): 45-50.
15. Setyarini, Anggun, and Heni Dwi Windarwati. “Influence Factors of Emergency Medical Services (EMS) Prehospital Time Interval Variety: A Systematic Review.” Jurnal Ners 15 (2020): 51-440.
16. Johns, G., V. Samuel, L. Freemantle and J. Lewis, et al. “The global prevalence of depression and anxiety among doctors during the covid-19 pandemic: Systematic review and meta-analysis.” Journal of affective disorders 298 (2022): 431-441.
17. Guolo, Annamaria, and Cristiano Varin. “Random effects meta-analysis: The number of studies matters.” Stat Methods Med Res 26 (2017): 1500- 1518.
18. Endalamaw, Aklilu, Nega Tezera Assimamaw, Tadesse Awoke Ayele, Achenef Asmamaw Muche, Ejigu Gebeye Zeleke, Amare Wondim, Getaneh Mulualem Belay et al. “Prevalence of childhood Cancer among children attending referral hospitals of outpatient Department in Ethiopia.” BMC cancer 21 (2021): 1-10.
19. Shamebo, M. “Acute leukaemias in adult Ethiopians in a teaching hospital.” Ethiop Med J 32 (1994): 17-25.
20. Enawgaw, Bamlaku, Melak Aynalem, Mulugeta Melku and Fikir Asrie, et al. “Hematological malignancies in the Northwest Ethiopia.” Plos one 16 (2021): e0260639.
21. Ayalew, Mulugeta. “PATTERN AND OUTCOMES OF CHILDHOOD MALIGNANCIES AT UNIVERSITY OF GONDAR HOSPITAL, ETHIOPIA: NONCONSECUTIVE CASE SERIES.” Ethiop J Ped Child Health 12 (2016).
22. Awedew, Atalel Fentahun, Zelalem Asefa, and Woldemariam Beka Belay. “National Burden and Trend of Cancer in Ethiopia, 2010–2019: a systemic analysis for Global burden of disease study.” Sci Rep12 (2022): 12736.
23. Ahirwar, D. R., R. K. Nigam, and D. Parmar. “A study of leukemias profile in central India.” Trop Plant Pathol 4 (2018): 2456-1487
24. Memirie, Solomon Tessema, Mahlet Kifle Habtemariam, Mathewos Asefa and Biniyam Tefera Deressa, et al. “Estimates of cancer incidence in Ethiopia in 2015 using population-based registry data.” J Glob Oncol 4 (2018): 1-11.
25. Ram, Ron, Anat Gafter‐Gvili, Liat Vidal and Mical Paul, et al. “Management of adult patients with acute lymphoblastic leukemia in first complete remission: Systematic review and meta‐analysis.” Cancer 116, no. 14 (2010): 3447-3457.
26. Ponvilawan, Ben, Pongthep Vittayawacharin, Pattaraporn Tunsing, and Weerapat Owattanapanich. “Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis.” TECHNOL CANCER RES T 20 (2021): 15330338211037434.
27. Owattanapanich, Weerapat, Pongprueth Rujirachun, Patompong Ungprasert and Jassada Buaboonnam, et al. “Prevalence and clinical outcome of Philadelphia like acute lymphoblastic leukemia: systematic review and meta-analysis.” Clin Lymphoma Myeloma Leuk 20 (2020): e22-e29.
28. Garniasih, Dina, Susi Susanah, Yunia Sribudiani, and Dany Hilmanto. “The incidence and mortality of childhood acute lymphoblastic leukemia in Indonesia: A systematic review and meta-analysis.” PLoS One 17 (2022): e0269706.
29. Radhakrishnan, Vivek S., Narendra Agrawal, Bhausaheb Bagal, and Ishan Patel. “Systematic review of the burden and treatment patterns of adult and adolescent acute lymphoblastic leukemia in India: comprehending the challenges in an emerging economy.” Clin Lymphoma Myeloma Leuk 21 (2021): e85-e98.
30. Gelibo, Terefe, Theodros Getachew, Abebe Bekele and Atkure Defar, et al. “Availability and readiness of services for cancer care at health facilities in Ethiopia: implication for action.” Ethiop J Health Dev 31 (2017): 391-396.
31. Force, Lisa M., Ibrahim Abdollahpour, Shailesh M. Advani and Dominic Agius, et al. “The global burden of childhood and adolescent cancer in 2017: An analysis of the Global Burden of Disease Study 2017.” The Lancet 20 (2019): 1211-1225.
32. Rodriguez-Galindo, Carlos, Paola Friedrich, Patricia Alcasabas and Federico Antillon, et al. “Toward the cure of all children with cancer through collaborative efforts: pediatric oncology as a global challenge.” Clin Oncol 33 (2015): 3065-3073.
33. Dong, Ying, Oumin Shi, Quanxiang Zeng and Xiaoqin Lu, Wei Wang, Yong Li, and Qi Wang. “Leukemia incidence trends at the global, regional, and national level between 1990 and 2017.” Experimental hematology & oncology 9 (2020): 1-11.
34. Lin, Xiangjie, Jinghan Wang, Xin Huang, Huafeng Wang, Fenglin Li, Wenle Ye, Shujuan Huang et al. “Global, regional, and national burdens of leukemia from 1990 to 2017: A systematic analysis of the global burden of disease 2017 study.” Aging (Albany NY) 13, no. 7 (2021): 10468.
35. IntHout, Joanna, John PA Ioannidis, and George F. Borm. “The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian Laird method.” BMC Med Res Methodol 14 (2014): 1-12.
36. Israe͏̈ls, Trijn. Aspects of the management of children with cancer in Malawi. Vol. 51. 2008. Getachew, Sefonias, Mirgissa Kaba, Muluken Gizaw, and Girma Taye. “Health service access, utilization and prevailing health problems in the urban vulnerable sections of Ethiopia.” Ethiop J Health Dev 34 (2020).