Gagné F1*, Roubeau-Dumont E1,2
1Aquatic Contaminants Research Division, Environment and Climate Change Canada, Montréal, Québec, Canada. H2Y2E7.
2Institut national de la recherche scientifique, Centre Eau Terre Environnement, Québec, Québec, Canada.
*Correspondence: Gagné F, Aquatic Contaminants Research Division, Environment and Climate Change Canada, 105 McGill, Montréal, Québec, Canada H2Y 2E7, E-mail: francois.gagne@ec.gc.ca
Received: 16 August 2024; Accepted: 30 December 2024; Published: 06 January 2025
Citation: Gagné, F, Smyth SA and André C. “Plastic Contamination and Water Quality Assessment of Urban Wastewaters.” J Environ Toxicol Res (2025): 111.
DOI:doi.org/10.59462/3068-3505.2.1.111
Copyright: © 2025 Gagné F. This is an open-ac cess article distributed under the terms of the Cre ative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Biofilms are usually found on rocky surfaces in streams, rivers and lakes, which are under the influence of multiple pollution types, including nanoplastics (NPs). The purpose of this study was to examine both the influence of anthropogenic activity (pollution) on the occurrence of NPs and the plasticizer dibutylphthalate (DBP) on the functional characteristics of biofilms. Biofilms were collected on rocks at different sites with a gradient of anthropogenic activities, and were analyzed for DBP and NPs using a plasmonic gold nanosensor. Their functional properties, such as proteins, lipids, viscosity, chlorophyl a/b, esterase, aldehydes (oxidative stress) and polyurethane biodegradation index (BDI), were assessed as well. The results revealed that biofilms downstream a large city and agricultural sites contained higher amounts of DBP and NPs. The BDI and esterase activity, involved in the degradation of plastic esters, were dampened at sites under anthropogenic pollution, suggesting a negative impacts on plastic biodegradation. The levels in viscosity, lipids and proteins were also affected in biofilms from anthropogenic sites, and showed signs of oxidative stress. It is concluded that biofilms at polluted sites are contaminated by plastic related compounds and display reduced capacity to degrade plastic esters.
Keywords
Nanogold plasmonic probes • Nano plastics • Biofilms • Biodegradation index • Oxidative stress
Introduction
Biofilms are a community of microorganisms composed of bacteria, cyanobacteria, and algae (diatoms) which are found in virtually all freshwater aquatic streams, rivers and lakes [1]. Biofilms composition and properties were shown to respond to many environmental contaminants involved in biodegradation of organic matter [2] including plastic polymers of interest for water quality monitoring. They excrete a complex array of polysaccharides, proteins and lipids forming a gel-like matrix (exudates) attached to miscellaneous surfaces (rocks, concrete, plastics, mussels and plants) in aquatic environments, which are called extracellular polymeric substances (EPS). These communities are sensitive their environments and nutrient status [3]. Exposure to phosphate in laboratory-reared biofilms increased the number of resistant taxa towards pollution. In contrast, exposure to Zn and/or Cd caused a marked reduction in community diversity in nutrient rich and organic pollution taxa. Besides changes in the community structure of biofilms, another response towards water quality changes consists in the production of EPS. For example, exposure of microalgae biofilms to Cd for 48h induced the synthesis of polysaccharides and proteins as a defence mechanism against cellular damage [4]. In another study, Pseudomonas aeruginosa stimulated the production of EPS when exposed to polystyrene nanoplastics (PSNPs) [5]. Bacteria from the Pseudomonas sp involved in the biodegradation of plastic ester, such as polyurethane and over-expressed esterase activities [6]. In another study, bacteria exposed to NPs and microplastics (MPs) and other contaminants (copper, cadmium and polyaromatic hydrocarbons) led to oxidative stress and cell membrane damages [7].
The environmental contamination by plastic debris represents a major and widespread pollution problem [8,9]. The production of synthetic plastics has raised exponentially since the 1950s, and was not adequately supported by safe and durable waste management efforts. Plastics materials ranging in size from mm to the µm (MPs) reaching nm (NPs) are found nearly everywhere nowadays, reaching the highest levels in areas close to anthropogenic activities such as city, roads, agriculture and industrial sectors. Both MPs and NPs represent another substrate for colonization by microorganisms (biofilms), which is an important aspect of the plastisphere [10]. Sludge-containing plastics were shown to reduce the settling and dispersion rates of solids, thereby remaining in the suspended matter fraction in the water column [11]. The biofilms could potentially degrade plastics and release plastic-derived chemicals (such as dibutylphthalates, styrene, bisphenol A), including other existing chemicals known to adsorb to the surface of plastics. The influence of NPs in biofilms composition is not well understood at the present times, let alone the influence of various anthropogenic activities.
The purpose of this study was to determine the relative levels of NPs in biofilms collected at various sites exhibiting different levels of anthropogenic activities, hence pollution, and to link exposure to potential differences in biofilms functional properties. Our hypothesis was that NPs are found at higher levels near anthropogenic source of pollution. In parallel, functional properties of biofilms such as esterase activity, biodegradation of polyurethane, viscosity, lipids and reactive oxygen species were be examined in relation to the presence of accumulated NPs.
Materials and methods
Biofilm sampling
Biofilms were collected in July 2023 by scrapping the top surface of 5 to10 rocks (4-8 cm length and width) with a silicone brush. A total of 125 mL of filtered mineral water (0.2 µm on glass fiber, 47 mm diameter) was used to rinse the biofilm in an aluminum foil pan, and the composite sample was then transferred into a glass container. Special care was taken to collect rocks in riffle/run reaches under similar lighting conditions to minimise variability in biofilms formation. Biofilms were kept in a cooler at 4oC until arrival at the laboratory, lyophilised and divided in 3 technical replicates per collecting site. Samples were then stored in the dark.
Analysis of dibutylphthalae and plastic nanoparticles
The biofilms were thawed at room temperature and 200 µL of sample was mixed with one volume (200 µL) of 5 M NaCl, followed by one volume of acetonitrile as previously described [12]. The mixture was vortexed for 30 sec and centrifuged 1 min at 1500 rpm to separate the phases. The upper acetonitrile fraction was analyzed for dibutylphthalate (DBP) and polystyrene nanoplastics (NPs) by a plasmonic nano-gold (nAu) sensor array. The presence of plastic nanoparticles was also analyzed based on the solvatochromic property of Nile red dye as previously described [13]. The determination of DBP was determined using arginine-coated nAu as previously described [14]. Citrate-coated 10 nm nAu nanoparticles at 50 mg/mL were purchased from Nanocomposix (USA). The nAu suspension was centrifuged at 20 000 x g for 10 min at 4oC, the pellet was resuspended in MilliQ water, and subsequently treated with 0.5% arginine for 3 h at room temperature. Another step of centrifugation (similar speed) allowed to remove excess and unbound arginine. The assay consisted in mixing 150 µL of arginine-nAu with 10 µL of the acetonitrile fraction, and 10 µL of HCl 0.1 M. The absorbance was then measured between 500-750 nm to determine changes between dissociated nAu (A 530 nm) and aggregated nAu (A 620 nm). For the determination of polystyrene nanoparticles concentrations (PsNPs), a plasmoic nAu assay was also used. The assay was developed initially for drinking water but was modified for more complex biological samples [15]. The assays eliminate most intereferences by a NaCl salting out step followed by the extraction of plastic nanoparticles with acetonitrile. Nanoplastics are then determined in the acetonitrile fraction and calibrated by the standard addition methodology. Briefly, the stock nAu suspension from the same supplier were incubated with 0.01% of mercaptoundecanoic acid (MUA) overnight [15,16], and centrifugated at 20 000 x g for 10 min with the pellet subsequently resuspended in MilliQ water to remove unbound excess MUA and citrate buffer. A sample volume of 10 µL of the acetonitrile fraction was mixed to 150 µL of MUA-nAu followed by the addition of 10 µl of 0.1 M HCl. The formation of NPs-MUA-nAu complex was determined by the appearance of a reddish color (absorbance at 570 nm) from the violet (aggregated) complex (absorbance at 620 nm) in the absence of PsNPs. Color analysis was achieved by either spectrometry (absorbance at 530, 570 and 620 nm) or by picture analysis using an RGB-color detection software (freeware from the web) from a standard smartphone equipped with a 5-megapixel resolution camera. The relative levels of plastic nanoparticles were expressed as ref (R)-channel color or A 570 nm increase/mg dry weights. Calibration was achieved with standards solutions (0.1, 0.5 and 2 µg/mL) of PsNPs (50 nm diameter, Polyscience, USA) in the presence of 10 % acetonitrile in MilliQ water. Previous studies determined that the nAu sensor could detect other types of plastics such as polyethylene terephthalate, polyethylene, and polypropylene/polyamide [16].
The solvatochromic properties of Nile red (NR) dye were also examined in the biofilms extracts in the attempt to detect plastic-like particles as previously described [13]. The fluorescence of NR depends on the polarity of its environment, where plastic polymers offer a less polar environment than lipids, leading to a blue shift in the NR emission spectra. A 10 µL sample of the biofilm extract was mixed with 190 µL of 10 µM NR dye (made in PBS: 140 mM NaCl, 2 mM KH2PO4 and 1 mM NaHCO3, pH 7.4). The mixture was analyzed by scanning fluorometry between 520-700 nm at an excitation wavelength of 485 nm (10 nm bandwith, Neo-2 Synergy, Bioteck Instrument, USA). The first derivative of the emission spectra followed by Fourier transformation curve fitting (the first 10 frequencies) were performed to remove the background noise of the instrument. Standards of PsNPs (50 nm) were used and emitted at 620 nm, compared to the major emission peak at 640-660 nm of the lipid environment in biofilms [13].
Biofilm functional properties
Biofilms (no NaCl addition/acetonitrile extraction) suspensions were allowed to settle in ice before determination of viscosity, total lipids/proteins, esterase activity, chlorophyll a and b (chla, chlb) and polyurethane degradation index (BDI). The BDI was devised to determine whether biofilms contaminated by pollution (plastics) could adapt to degrade plastic materials. For viscosity, 20 µL of suspension was mixed with 10 µM (9-(dicyanovinyl)-julolidine) and fluorescence determined at 450 nm excitation/520 nm emission [17]. Standard solutions of glycerol (0.1-1 %) was used for calibration and the data were expressed as relative fluorescence units (RFU)/mg dry weight biofilms. Total proteins were determined using the SDS methodology. Briefly, 10 µL of the suspensions was mixed with 40 µL of 0.1% SDS for 5 min, heated at 60oC for 10 min and volume completed to 200 µL in PBS. The absorbance was measured at 280 nm (aromatic amino acids) using a microplate reader (Neo-2 Synergy, Bioteck Instruments, USA), using standard solutions of albumin for calibration. The data was expressed as mg proteins/mg dry weight biofilms. Total lipids were determined using the NR dye as previously described [18]. A 50 µL sample was mixed with 150 µL of 10 µM NR in PBS and fluorescence was observed at 485 nm excitation/650 nm emission. Standard solutions of Tween-20 were used for calibration and the data were expressed as RFU/mg dry weight biofilms. The levels of aldehydes, a marker of oxidation of proteins, were determined using the 4-aminofluorescein methodology [19]. A 20 µL sample of the suspension was mixed with 180 µL of 10 µM 4-aminofluorescein in PBS, and fluorescence determined at 485 nm excitation/530 nm emission in 96-well dark microplates. The data were expressed as RFU/mg dry weights biofilms. The levels of total chlorophyll a and b were determined by fluorescence as previously described [20] (in biofilm samples diluted 1/10 in PBS. Chlorophyll a (more abundant in cyanobacteria) and b (found in most algae/plants) were determined at 425 nm excitation/680 nm emission and 485 nm excitation/600 nm emission respectively in dark microplates (Neo-2 Synergy, Bioteck Instruments, USA). The data were expressed as RFU/mg dry weight biofilms. Esterase activity was determined using the p-nitrophenyl acetate substrate at absorbance at 412 nm [21].
Polyurethane degradation activity
The capacity of biofilms extracts to degrade polyurethane a common source of plastic related materials in the environment, were determined in vitro following a previously published procedure using a different source of polyurethane polymers [6]. A 50 µL sample of biofilm suspensions was mixed with 10% polyurethane diol (Sigma Chemical Company, On, Canada) in PBS, and the refractive index (RI) measured (t0). The RI was determined using MilliQ water as blank (RI=1.3316) and 10% polyurethane (RI=1.3352 at t0) using an electronic RI device (HDR-P5, Fisher brand, Canada). The mixture was allowed to stand for 12 h at 20oC in the dark and the RI was measured again (t12h). The decrease in the RI determined the breakdown of polyurethane polymers and constitutes a biodegradation index. The data were expressed as the biodegradation index: decrease in refractive index (t0- t12h)/mg dry weight biofilms.
Data analysis
In this study, biofilm samples were collected at 16 sites, which were grouped as reference (REF), rural (Rural), Agriculture (Agri), upstream city area (Ups-city), downstream city area (Downs-city) and a commercial harbor (Harbor) as depicted in (Table 1). The general map of the sites is shown in the supplementary material section (Figure 1S). The data were expressed as the mean with the standard error and analysed using non-parametric analysis of variance (ANOVA). Significant differences between the reference and the other sites were determined using the Conover-Iman test. Correlation analysis was performed using the least square procedure. Significance was set at p < 0.05 and the tests were performed using the Statsoft software package (version 13, USA).
Results
Nanoplastics and DBP contents
The sites from REF group consisted of streams located upstream populated area with no apparent source of human activity (forest) in the area. Sites from the Rural group were relatively close to anthropogenic activity (between small townships and/or field crops with roads). The data revealed that the Downs-city and Agri groups contained higher levels of NPs compared to the reference sites, using the plasmonic nAu sensor (Figure 1). The harbor, rural and a downs-small city seemingly had higher levels of NPs, but were not significantly different from the REF group. Similarly as for NPs, DBP levels were significantly increased at the Agri and both the Downs city sites (from large and small cities) compared to the reference site (Figure 1). The Ups-city site had lower levels of DBP relative to the REF group. Correlation analysis revealed that NPs levels were correlated with DBP levels (r=0.78), (Table 2). The presence of plastic materials was assessed using the solvatochromic properties of NR dye in biofilm extracts (Figure 2). The emission spectra of NR in the presence of biofilm extracts revealed the usual lipid peak at 650 nm. A blue shift in the emission peak was observed in samples compared to the REF group (Figure 2A). In the attempt to better resolve the emission spectra, the first derivative of the emission spectra was calculated and fitted using Fourier transformation (Figure 2B). The data revealed the appearance of a peak at 620 nm consistent with plastic-like compounds (polystyrene nanoparticles) at some sites such as Downs-city centre and the harbor groups. Given that this approach was rather used to qualitatively detect PsNPs than provide quantitative data in the present study, the presence of less-polar compounds (620 nm emission) is consistent with the presence of polystyrene plastics or other compounds sharing similar hydrophobicity, in respect to NR. The levels of plastic-like materials based on the blue shift in NR fluorescence at 620 nm were significantly elevated at the Agri and Downs large city groups. Correlation analysis revealed that the shift in NR was significantly correlated with NPs (r=0.4) but not with DBP levels in biofilms.
Biofilms contamination and functional properties
The capacity of biofilms extracts to degrade polyurethane diol polymer was examined using the BDI (Figure 3). The analysis revealed that the BDI was generally lower than the REF sites where the lowest BDI was at the harbor sites. The BDI was not significantly correlated with either NPs or DBP levels. This suggests that biofilms from polluted sites generally have lower capacity to degrade polyurethane by multiple contamination. The activity in carboxylesterase was determined in biofilms across the groups (Figure 4) and followed the same pattern as the BDI. Esterase activity was globally and significantly lower in all the groups compared to the REF group, and correlation analysis did not show a significant link between esterase activity with the above endpoints. This indicates that biofilms from polluted area display lower capacity to biodegrade polyurethane (a proxy for plastics) and esterase activity involved in lipid hydrolysis and plastic esters. The levels of aldehydes, a marker of oxidative stress, were significantly increased and decreased at the Agri and the Ups-city groups, respectively (Figure 4). Oxidative stress (aldehydes) was correlated with DBP (r=0.45) and NPs (r=0.35) levels. The levels of lipids and proteins in biofilms were also determined (Figure 4). The levels of lipids were significantly lower at the Ups-city and higher at the Agri groups (P-values). Lipids were significantly correlated with NR shift (r=0.49), NPs (r=0.56), DBP (r=0.63) and proteins (r=-0.47). The levels in proteins were significantly lower at polluted sites such as harbor and the Downs (large and small) city groups (P-values). Total proteins in the biofilms were correlated with NPs (r=-0.41), DBP (r=-0.55) and esterase activity (r=0.36) and aldehydes (r=-0.63).
The changes in viscosity and total chlorophyll a and b were determined in the biofilms (Figure 5). Viscosity was significantly lower and higher at the upstream city and agriculture groups, respectively (P-values, Figure 5). Correlation analysis revealed that viscosity in biofilm extracts was related to the NR shift (r=0.36), NPs (r=0.39), DBP (r=0.7), aldehydes (r=0.36), proteins (r=-0.6) and lipids (r=0.69). The levels of chlorophyll a (more abundant in cyanobacteria) and b (green algae and plants origin) were also determined (Figure 5). In respect to the REF group, chl a and b were significantly higher at the agri, rural and downs-city groups, with a trend in higher chl a levels compared to chl b. In the other groups, chl b levels were Correlation analysis revealed that chl a levels were significantly correlated with viscosity (r=0.54), NR emission shift (r=0.44), lipids (r=0.67), chl b (r=0.81) and DBP levels (r=0.32). Chl b levels were correlated with DBP (r=0.4), proteins (r=-0.41), NPs (r=0.36), viscosity (r=0.75), NR shift (r=0.49) and lipids (r=0.69). In the attempt to gain a more global understanding of the biofilm properties, a principal component analysis was performed (Figure 6). The analysis revealed that the following groups had the highest factorial scores: Agri, Downs-city (large and small) and rural, with 45% of the variance explained by one factor. The most important biomarkers, based on factor coefficients, were as follows: viscosity~lipids>Chl b~DBP~chl a~NPs~proteins.
| Sites | Coordinates | Groups | Annotation |
| (figures) | |||
| Île aux foins | 46.024188, -73.180070 | Rural | Rural |
| Île du milieu | 46.050885, -73.179695 | Rural | Rural |
| St-Ignace-de-Loyola | 46.055768, -73.157048 | Upstream-City | Ups-city |
| St-Ignace-de-Loyola | 46.064772, -73.112587 | Downstream-City (pop. 2500) | Downs-city |
| Île des Barques | 46.089245, -72.984235 | 12 km Downstream-City | Downs-city-large |
| (Sorel; pop. 40 000) | |||
| Baie maskinongé | 46.179758, -72.972755 | Rural | Rural |
| Yamachiche | 46.261333, -72.825950 | Rural | Rural |
| Baie du Febvre | 46.202550, -72.724933 | Reference | Ref |
| Pierreville | 46.133733, -72.857917 | Agriculture | Agri |
| Île à la pierre | 46.078133, -73.031250 | 8 km Downstream-City | Downs-city-large |
| (pop. 40 000) | |||
| Île aux sables | 46.123967, -72.968100 | 16km Downstream-city | Downs-city-large |
| (pop. 40 000) | |||
| Baie de girodeau | 46.146750, -72.992450 | Rural (no direct contact with agriculture field) | Rural |
| Grande île | 46.118567, -73.039450 | Reference, north shore of Saint-Lawrence (between islands) | Ref |
| Port de Sorel | 46.049417, -73.125900 | Harbor with commercial and touristic boat traffic | Harbor |
| 3 lakes stream | 46.986552, -71.391944 | Rural, not directly in line with villages/cities | Ref |
| Jacques-Cartier | 47.070488, -71.426826 | Rural, not directly in line with villages/cities | Ref |
Table 1. Site identification and categorization
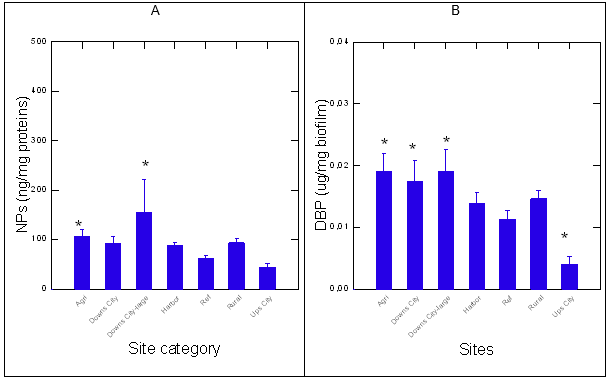
Note: The data represent the mean with standard error. The star * symbol indicates significance from the Ref site.
Figure 1. Levels of nanoplastic particles and DBP in biofilms.
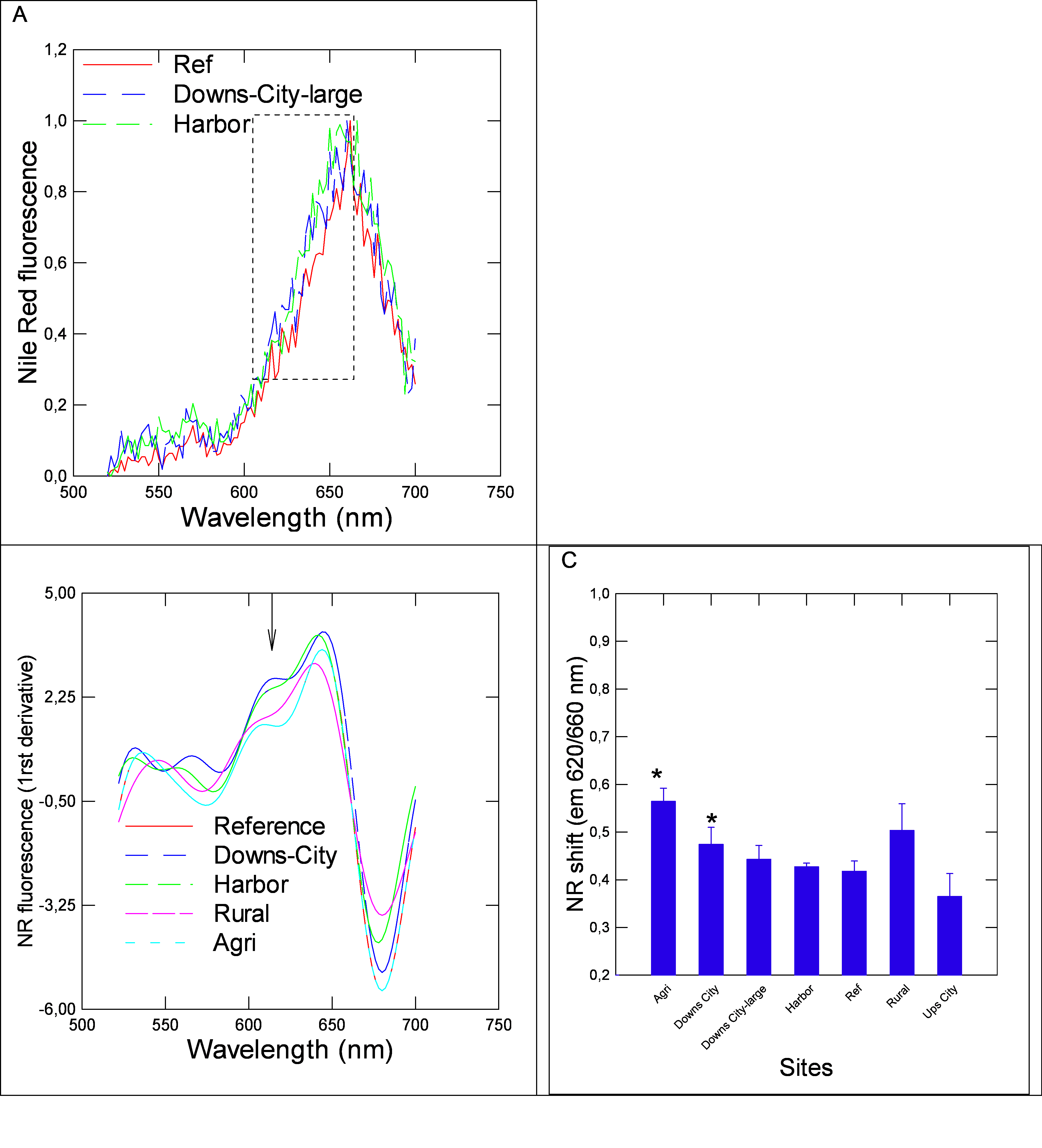 Figure 2.Solvatochromic analysis of Nile red fluorescence in biofilm extracts. higher than chl a, but the trend wasn’t significantly different from the REF group.
Figure 2.Solvatochromic analysis of Nile red fluorescence in biofilm extracts. higher than chl a, but the trend wasn’t significantly different from the REF group.
Abbreviations: Proteins (Pro), Nanoplastics (NPs), viscosity (Visc), Neutral red shift at 620 nm (NR(Sol)), biodegradation index (BDI), esterase (Est), chlorophyll a (Chla), chlorophyll b (Chlb), aldehydes (Ald), dibutylphthalate (DBP). Significant correlations are found in bold (r>0.3).
| Pro | NPs | Visc | NR shift | BDI | Lipids | Est | Chla | Chlb | Ald | DBP | |
| Pro | 1 | ||||||||||
| NPs | -0.42 | 1 | |||||||||
| Visc | -0.6 | 0.39 | 1 | ||||||||
| NR shift | -0.21 | 0.4 | 0.36 | 1 | |||||||
| BDI | -0.01 | -0.13 | -0.04 | -0.02 | 1 | ||||||
| Lipids | -0.47 | 0.56 | 0.69 | 0.5 | 0.01 | 1 | |||||
| Est | 0.37 | -0.26 | -0.25 | 0.1 | 0.13 | -0.15 | 1 | ||||
| Chla | -0.11 | 0.24 | 0.54 | 0.44 | 0.04 | 0.67 | -0.07 | 1 | |||
| Chlb | -0.41 | 0.36 | 0.75 | 0.49 | -0.07 | 0.69 | -0.22 | 0.81 | 1 | ||
| Ald | -0.63 | 0.35 | 0.36 | -0.11 | -0.03 | 0.1 | -0.3 | -0.27 | -0.1 | 1 | |
| DBP | 0.56 | 0.78 | 0.7 | 0.22 | 0.04 | 0.63 | -0.04 | 0.32 | 0.4 | 0.45 | 1 |
Table 2. Correlation analysis of biofilms properties
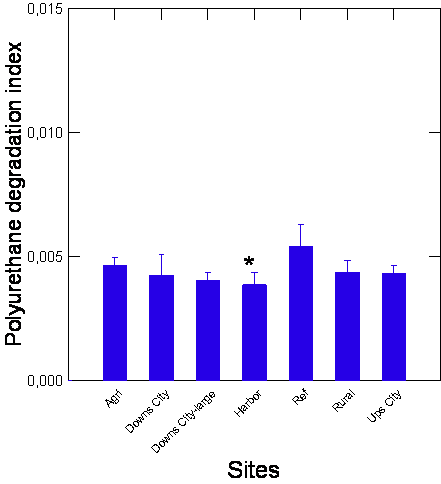
Note: Biofilm S2 fractions were incubated with 10% polyurethane for 24h at 20oC and the loss of polyurethane were determined by the refractive index. The data are expresses as the mean with standard error. The star symbol indicates significance at p=0.05 compared to the reference site.
Figure 3. Polyurethane degradation index in biofilm fractions.
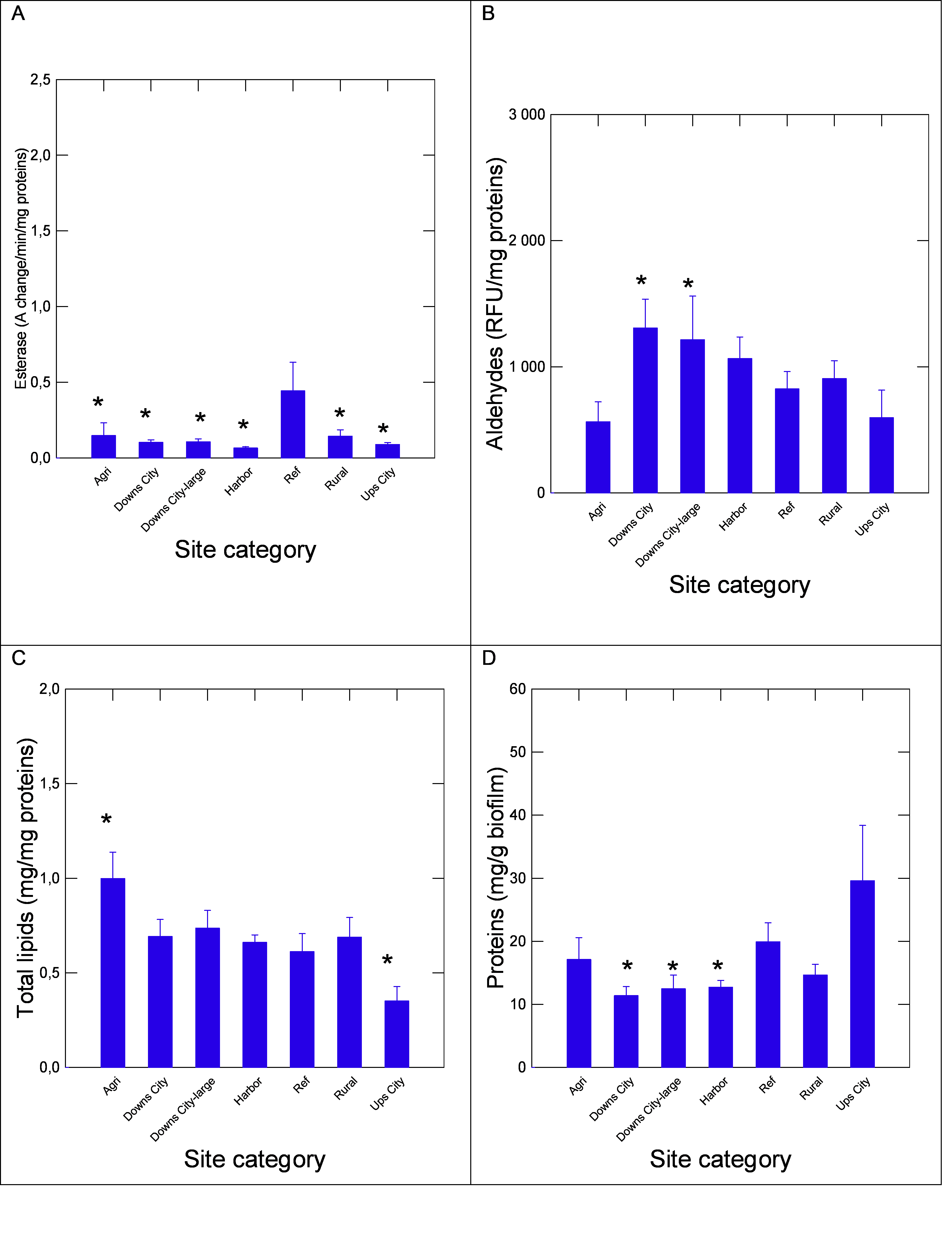
Note: Metabolic activity in biofilms fractions was determined by following changes in proteins (A), total lipids (B), esterase activity (C) and oxidative stress by aldehydes content (D). The data represent the mean with the standard error. The star symbol * indicates significance from the reference sites.
Figure 4. Energy components and metabolic activity biofilms
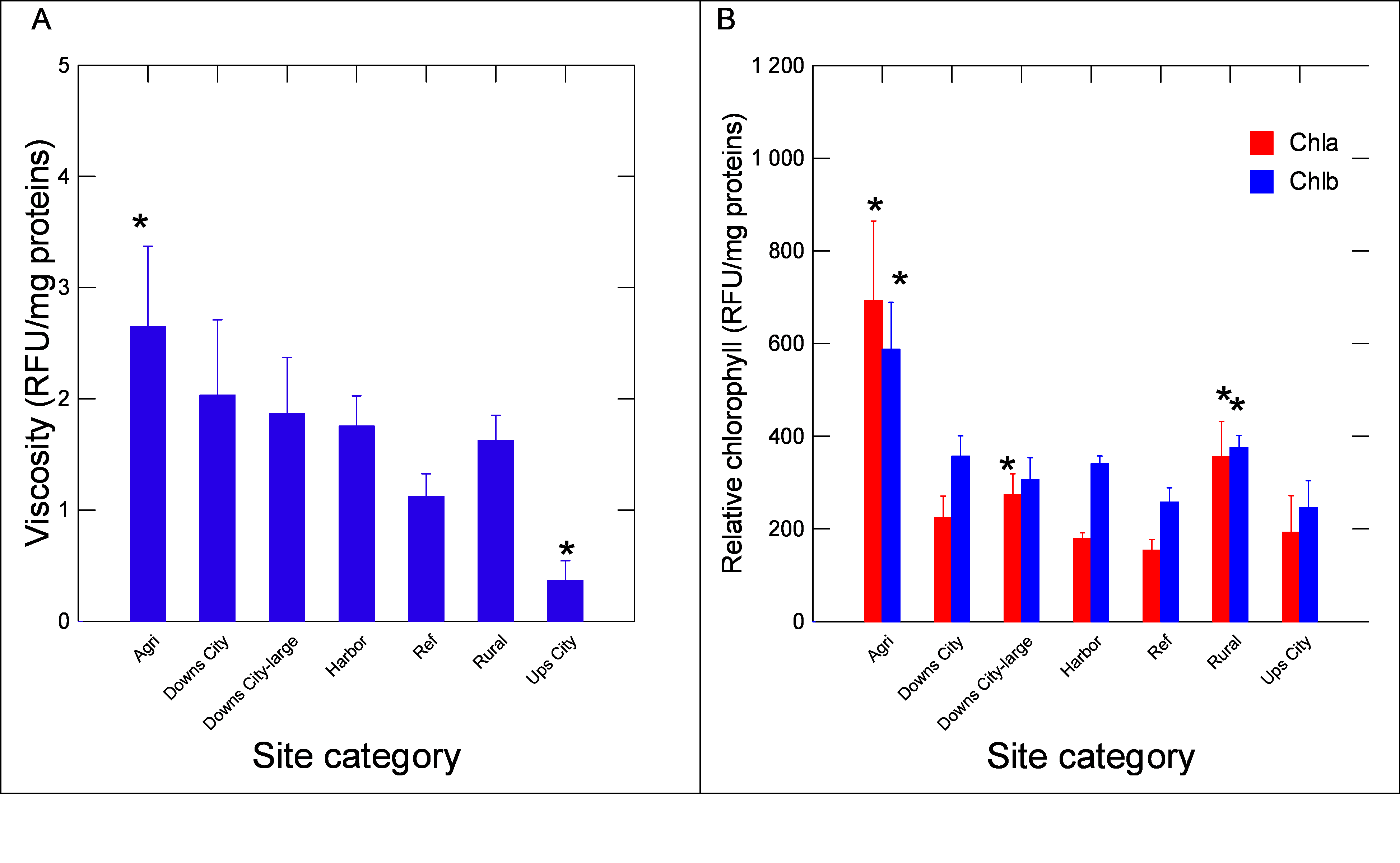
Note: The changes in viscosity (A) and chlorophyll ab (B) were determined in biofilms. The data represent the mean with the standard error. The star symbol * indicates significance from the reference sites.
Figure 5. Changes in viscosity and chlorophyll contents in biofilms.
Discussion
Although plastic contamination, as determined by DBP and PsNPs in biofilms, tended to increase at sites near anthropogenic activity, the metabolic activity (esterase) and BDI were not increased suggesting that pollution reduced the biofilms metabolic activity and their ability to degrade plastic esters (polyurethane). The BDI used polyurethane, which is a plastic polymer usually associated with melamine in thermoplastics materials (kitchen and toilet counters etc). In this study, a general decrease in esterase activity and BDI was found in biofilm extracts, which is consistent with the involvement of esterase activity in the degradation of polyurethane by Pseudomonas sp [6]. Indeed, polyurethane is an ester polymer of urethanes, which could be hydrolyzed by non-specific esterases in microorganisms. The presence of organics phthalates and esters found in dust in urban area were shown to reduce bacteria growth and increased sensitivity to oxidative stress in three opportunistic bacterial species (Escherichia coli, Enterococcus faecalis, and Pseudomonas aeruginosa [22]. This effect was found in nutrient-rich conditions; in the absence of nutrients, dust could potentially increase growth through adaptation to different carbon sources. In biofilm producing Salmonella enterica, growth was reduced by exposure to chromium, cadmium and copper [23]. The lipophilic plastic contaminant, DBP with its log Kow = 5.6, would then accumulate in lipid-rich environments with bioaccumulation factor in the order of 3000 [24]. Reduced growth in Karenia brevis was observed at the 10 mg/L range for phthalate esters and that toxicity could arise for environmental concentrations as low as 3 µg/L [25]. The levels of DBP in biofilms reached values from below detection limit up to 25 µg/g biofilms, suggesting that DBP could partially contribute to reduced biofilm esterase activity and BDI. In another study, adverse effects of DBP on Legionella pneumophila were found at 0.28 mg/L, suggesting that DBP could alter the function of bacteria/algae in biofilms [26]. The levels of DBP reached 0.38 µg/L in some water bodies in China [27] and considering the bioaccumulation factor for DBP, these concentrations can reach toxic levels for bacteria and algae. This corroborates the observed correlations between DBP levels in biofilms namely with chl b (r=0.4). This suggests that the algal fractions of the biofilms could be responsible, in part at least, with DBP levels found in biofilms. The influence of algal blooms in biofilms composition and function should be examined more closely in relation to plastic degration in the future. In the present study, DBP levels were significantly correlated with Chl a (cyanobacteria enrichment) and Chl b (algae) suggesting that DBP was perhaps more closely related to cyanobacteria, which are abundant in nutrient rich (polluted) environments. Interestingly, it has been shown that DBP and other phthalate were synthetized by algae and cyanobacteria [28]. The mean value of DBP in both algae and cyanobacteria was 46 ug/g dry weight algae, which is in the same range found in biofilms (not only composed of algae) in the present study (14 µg/g dry wet biofilm). This suggests that DBP found in biofilms is not solely of anthropogenic origin since a portion of the phthalate could have been biogenically produced. The levels of DBP were more strongly correlated with Chla (cyanobacteria) suggesting a nutrient-rich (eutrophic) environment such as agricultural and rural sites. However, the levels of DBP were significantly correlated with NPs (r=0.78) as determined by the nAu sensor probe, suggesting an anthropogenic origin of contamination. This was corroborated by an analysis of covariance for DBP levels corrected against NPs, showing lack of significance between sites i.e., the “natural” production of DBP were not significantly different between sites.
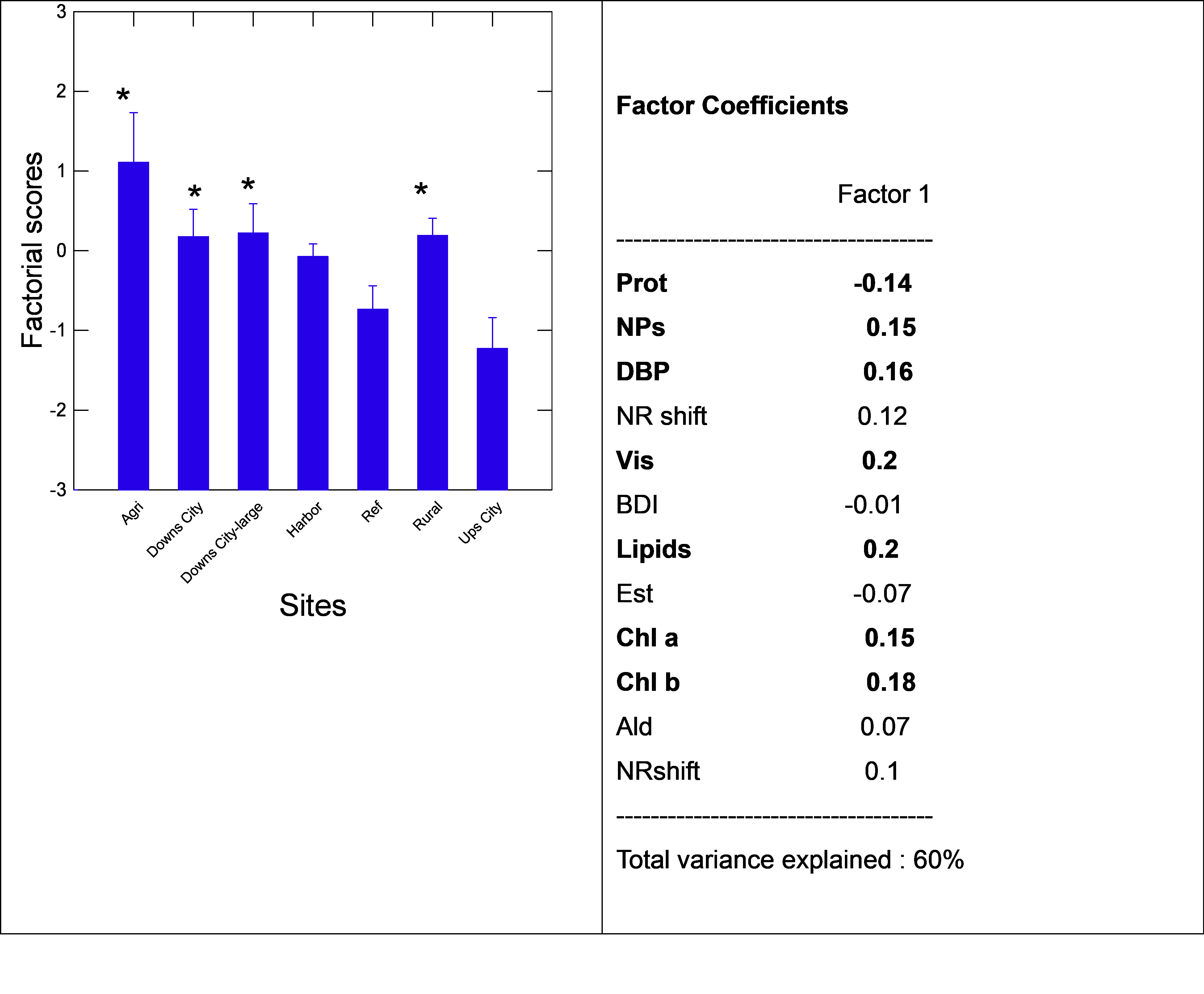
Note: Principal component analysis was performed on the various metrics of biofilms. The total variance was explained at 60 % and the following were the most important ones: viscosity, lipids, Chlb, DBP, NPs and proteins. Sites with the highest factorial scores were the Agri, rural and Downs city sites. Supplementary materials
Figure 6. Factorial components of biofilms
Biofilms produce an extracellular matrix, which can interact with plastic micro and nanomaterials [29]. It is mainly composed of polysaccharides followed by proteins, and therefore contains many functional groups, such as carboxyls, hydroxyls and amides. These functional groups can not only interact with weathered plastic particles, but also other contaminants that are carried by plastic particles (vector effect), such as metals, organics components including tire wear substances (such as hexamethoxymethylmelamine and 1,3-diphenylguanidine).
However, the interaction of plastics with the extracellular polymeric substances in biofilms favor further degradation and mineralization by the microflora. The density of biofilms (including the extracellular polymeric matrix) could be assessed by changes in viscosity on biomass basis. In this study, viscosity was generally higher (especially at agricultural sites) at sites under anthropogenic activity, although the upstream sites were lower than the ref site. Viscosity was also significantly correlated with DBP (r=0.7) and NPs (r=0.7) levels, corroborating that the contaminants are found in this extracellular matrix. It has been shown that cadmium exposure of algae-based biofilms increased proteins in the extracellular matrix, which is considered a stress response to this contaminant [4]. Viscosity was strongly correlated with proteins (r=-0.6), Chlb (r=0.75) and lipids (r=0.69) suggesting that lipids was also included in this extracellular matrix, although they are usually present at low concentrations. Indeed, the accumulation of lipids in flocculating/floating microalgae reached 27% dry mass following a 6 days exposure to municipal effluents [30], highlighting the affinity of biofilms towards lipids. These findings are consistent with the strong correlations between lipids and Chla (r=0.67) and Chlb (r=0.69) found in the present study (Table 2). An analysis of covariance of lipids against sites and esterase activity revealed that sites choice had a more significant influence (p<0.001) than esterase activity (p=0.02) on lipid levels in biofilms. This suggests that lipids found in biofilms depends less on the intrinsic esterase activity (metabolic activity) compared to local sources of lipids, such as environments near wastewaters disposal sites (municipal effluents) or rich in manure (Agricultural sites). Although it could be expected that plastics are found at higher concentrations at sites downstream urban/cities, the higher levels of plastic at sites/streams nearby agricultural activities were unexpected. The sources of plastic contamination are diverse and could result from soil/manure (organic fertilizers), application of pesticides in fields. This should be more closely examined since food products could represent the causal link between environmental plastics and health issues in the human population. Biosolids are included in organic fertilizers, which are known to accumulate plastic materials [31]. Although not destined for agriculture, solid electronic-waste and leachates could represent a source of plastic contamination in nearby fields [32]. The contamination of farmland by micro/nanoplastics also occurs by the contamination of the water irrigation systems [33]. The abundance of microplastics in the farmland soil ranged from 1800 to 87000 plastic items/ kg soils, with polyethylene as the predominant polymer with sizes below 180 µm. This highlights the importance of integrating urban management with agronomic practices to alleviate the plastic contamination problems. In conclusion, biofilms collected close to anthropogenic sites contained elevated levels of the plasticizer DBP and PsNPs. The levels of NPs and DBP were especially high downstream a large city and agricultural sites. Notwithstanding this, the capacity of biofilms to degrade plastic ester such as polyurethane were generally dampened by anthropogenic activity as with esterase activity, which is involved in plastic degradation. This suggests a negative (cumulative) impact of pollution to degrade plastics. Moreover, changes in viscosity, proteins, chlorophyll and lipids in biofilms found close to sites polluted by anthropogenic activities suggests that the EPS might be impacted as well, as suggested by the strong correlation of DBP levels and viscosity in biofilms. The data supports the hypothesis that biofilms are negatively affected by plastics in polluted environments and warrants further investigations on the cumulative impacts of pollution on biofilm integrity, which are the primary decomposers in aquatic ecosystems.
Acknowledgement
This work was funded by the Saint-Lawrence Action plan of Environment and Climate Change Canada and the Natural Sciences and Engineering Research Council of Canada on microplastics at INRS-Environment, Québec, Canada. The authors thank Prof Isabelle Lavoie and Lindsey Yvette Mouatcho from the National Scientific Research Institute (Sainte-Foy, Quebec, Canada) for biofilm collection in this study. Biodegradation.” Sci. Total Environ. 886 (2023): 163932.
Supplementary materials
Note: The biofilm collection area were generally located upstream the Lake Saint-Pierre between Sorel and Trois Rivières (Québec, Canada). The biofilms were collected on periphytons (aquatic plants). Some of the collection sites are mentioned in the figure. The exact location of each collection sites was provided with the GPS coordinates in Table 1.
Figure 1S. General location of the study.
References