Dr. Rohina Aggarwal1*, Dr Sumesh Choudhary2, Dr Kunur Shah3
1Professor and Head, Department of Obstetrics and Gynaecology, Institute of Kidney Diseases & Research Centre (IKDRC), Ahmedabad, India
2Institute of Kidney Disease and Research Centre
3Institute of Kidney Disease and Research Centre, Ahmedabad
*Correspondence: Dr. Rohina Aggarwal, Professor and Head, Department of Obstetrics and Gynaecology, Institute of Kidney Diseases & Research Centre (IKDRC), Institute of Transplantation Sciences (ITS), Ahmedabad, India, Email: drrohinaaggarwal@gmail.com
Received: 01 Dec, 2025; Accepted: 22 Dec, 2025; Published: 29 Dec, 2025.
Citation: Rohina Aggarwal, Sumesh C, and Kunur S. “Autoimmune Hepatitis and Outcome of Pregnancy: Two Case Reports.” J Gynecol Matern Health (2025): 124. DOI: 10.59462/3068-3696.3.2.124
Copyright: © 2025 Rohina Aggarwal. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Autoimmune hepatitis (AIH) is a chronic inflammatory liver disorder usually characterized by circulating autoantibodies and elevated serum globulin levels. The disease’s pathology is that of a gradual hepatocellular inflammation that cannot be explained by more common aetiologies of chronic liver disease, such as alcohol consumption, viral infection, exposure to hepatotoxic substances, and hereditary liver disorders. After recent advances in immunosuppressive treatment and increased clinical experience on AIH, pregnancies in this group of patient have been reported. There is a high morbidity and mortality risk for both foetus and mother from conception to the end of the puerperium. Control of innate and humoral immunity together with hepatic functions is critical, as is the necessity of prevention of “endothelial injury/inflammatory processes” and thrombotic events. In this article we are reporting two such cases with successful maternal -foetal outcome despite high risk status (one with cirrhosis, one with complex congenital heart disease).
Keywords: High-risk Pregnancy, Liver Cirrhosis, CaseReport
Introduction
The diagnosis of autoimmune hepatitis is similar in both pregnant women and the general population on the basis of serologic and histologic findings as well as the ex clusion of other types of chronic liver disease. [1] For in dividuals with consistent clinical or laboratory features, it is recommended to obtain serum anti-nuclear (ANA), an ti-smooth muscle (ASMA), anti-mitochondrial (AMA), and anti-liver/kidney microsomal-1 (ALKM-1) antibodies, as well as either an immunoglobulin G (IgG) or gamma globu lin level [2]. It is recommended for those who are nega tive for these autoantibodies to obtain anti-soluble liver antigen/liver pancreas antibody, anti-actin antibodies, and atypical perinuclear antineutrophil cytoplasmic (p-ANCA) antibodies. Often, the disease can be strongly suspected based on clinical and laboratory features. Approximately 20percent of individuals presenting with AIH features lack circulating ANA, ASMA, or ALKM-1 antibodies [3] but there is therapeutic response to anti-inflammatory therapy in such cases. Histologic evaluation is especially useful as part of the diagnostic evaluation for those with few or atyp ical findings, negative autoantibodies, and/or normal IgG levels.
Autoimmune hepatitis (AIH) is a rare chronic inflammatory disease with a low incidence (1/100 000) in the general population [4]. AIH is more frequent in females, and good pregnancy outcomes are reported in a limited number of clinical studies [5].
Patients with autoimmune disorders and positive autoanti body are at risk for obstetric complications and perinatal morbidity and mortality [6]. Women in reproductive age group with autoimmune disorders are now able to get pregnant and give birth under successful medical management due to advancement in this field [7]. The progress of the disease during pregnancy is unpredictable and in formation in the current literature is not conclusive. Some authors have described worsening of the condition as a potential consequence after conception [8]. While, others have reported regression or an unalterable state during pregnancy [9].
We had two cases one had relapse of disease in the ante natal period of pregnancy and the other patient who was in remission had uneventful with no relapse in antenatal period [10].
Case 1
Known case of Chronic Liver Disease-Overlap Syndrome (Auto Immune Hepatitis/Primary biliary Cholangitis) pre dominantly Auto immune hepatitis diagnosed since 2016 when she had fever, diarrhoea with deranged Liver Func tion Tests with rise in Serum Glutamic-Oxaloacetic Transa minase and Serum Glutamic Pyruvic Transaminase (SGOT and SGPT) levels along with normal Serum Bilirubin level. At that time, she had no pruritus, alopecia, oral ulcer sicca symptoms, arthralgia, raynaud’s phenomenon, paraesthe sia, rales, cough or dyspnoea, weight loss, photosensitiv ity or no bood in stools. She was diagnosed with Gluten Sensitivity and started on Gluten free diet.
Reports:
| Antinuclear Antibody (ANA)` | 4 Positive with coarse spectrum |
| Anti RO | Positive |
| Anti Endomysial Antibody (EMA -Ig A) | Positive |
| Anti-ds DNA | 61 |
| Anti-Mitochondrial Antibody (AMA) | Negative |
| Anti-Smooth Muscle Antibody (Anti SMA) | Negative |
| Liver Kidney Microsomal Antibody (LKM) | Negative |
| Antibodies to Tissue Transglutaminase (ANTI TTG12) | Negative |
| Duodenal Biopsy (2016) | Mild Villous Blunting |
| Liver Biopsy (2019) | Interphase Hepatitis with Cirrhosis (Auto Immune) |
| Fibro Scan Liver | LSM 13.5kpa,18.7kpa, 23.5kpa |
| USG Abdo0.men | Subtle coarse echotexture of liver with splenomegaly |
Table 1: Laboratory and Diagnostic Findings for Case 1 Patient (Chronic Liver Disease-Overlap Syndrome)
FibroScans
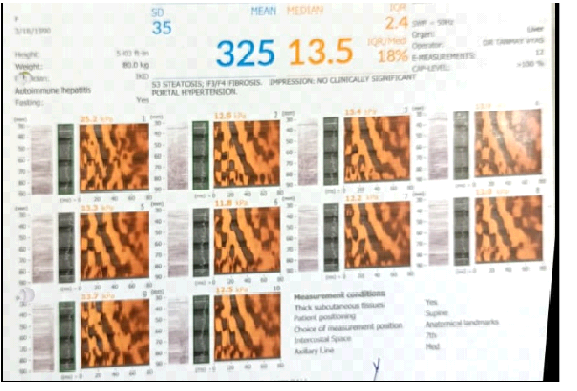
Figure 1: Baseline transient elastography

Figure 2: Follow‑up transient elastography
FibroScan is a quick, painless, non-invasive ultrasound based test to assess liver stiffness and fat content without a biopsy.
Fibroscans showing progressive increase in liver scar ring and fibrosis measured as rise in stiffness score from 13.5kPa to 18.7kPa to 23.5kPa
SLICC Criteria for SLE was not satisfied.
Final diagnosis was chronic liver disease with compen sated liver cirrhosis and AIH under remission.
A 28-year-old spontaneously conceived pregnant woman (Gravida 2, Para 0, Abortion1) Rh negative pregnancy with cirrhosis secondary to AIH was under regular follow up with us. She had an abortion at 8 weeks gestation in her previous pregnancy. In this pregnancy at 32wks, on Ultra- Sonography (USG) symmetrical intrauterine growth retardation (IUGR) with growth parameters of 29 weeks was detected. Indirect Coombs Test (ICT) was negative throughout the pregnancy. Glucose Tolerance Test (GTT) and Thyroid Stimulating Hormone (TSH) levels were nor mal. International Normalized Ratio (INR) was maintaining 1.1and Serum Albumin was between 2.5-2.9g/dL throughout the pregnancy.2D- Echocardiography revealed Ejec tion Fraction 60%, concentric left ventricular hypertrophy, reduced left ventricle compliance and mild MR. She deliv ered a live 2.3 KG female baby at 35+5wks. pregnancy by LSCS. Postpartum period was uneventful.
She conceived again in June 2021 spontaneously(G 3P1A1Live1). She had relapse of disease at 8wks of preg nancy with rise in Total Serum Bilirubin 2.7mg/dL and Dir ect Serum Bilirubin 1.2 mg/dL and Indirect Serum Bilirubin 1.5 mg/dL with deranged liver enzymes SGOT 129U/L, SGPT 48U/L and INR being 1.56. Hence her Prednisolone dose was increased to 20 mg OD from 10 mg OD and Azathioprine continued as 50 mg OD. After 2 wks. of treatment her lab parameters improved. Total Serum Bilirubin fall down to 1.2 mg/dL with Direct Serum Bilirubin 0.78 mg/ dL and Indirect Serum Bilirubin 0.42 mg/dL levels along with improvement in transaminases levels. SGOT being 38U/L, SGPT 26 U/L and INR 1.2.
At 36+2 weeks she was admitted with us. At that time, she was on Tab Azathioprine 50mg OD and Tab Prednisolone 10 mg OD for liver problem and consulting a hepatologist regularly and was on her antenatal care follow-up visits with us and all the necessary laboratory tests were performed showed normal biochemistry profile. Total Serum Bilirubin was 0.8 mg/dL, Direct Serum Bilirubin 0.4 mg/ dL and Indirect Serum Bilirubin 0.4 mg/dL. Liver enzymes were SGOT 34 U/L, SGPT 28 U/L AND INR 1.43, Total serum proteins 5.7 g/dL and with Serum Albumin was low with value of 2.0 g/dL and Globulin 3.7 g/dL and. All viral markers were negative. GTT and TSH were normal.
The patient was delivered by caesarean section at 36+4 week after giving Inj Betamethasone 12 mg IM 2dosees 24 hours apart for foetal lung maturity. Inj Albmin 20% and Inj Plasmolyte were administered for 2 days prior to CaesareanSection. A live born 2.035 kg male baby with APGAR Score of 5 was delivered and was kept along mother side.
Intraoperative Period-During Caesarean Section there was mild atonic post-partum haemorrhage which was managed with bimanual massage, 40 units Oxytocin drip in 500ml of Normal Saline, intramyometrial Injection of PGF2α and per rectal administration of Tab Misoprostol 800 μg.
Postoperative Period- Haemoglobin was 8.1gm% with normal Iron Profile along with massive pitting pedal oedema till shins. For that 2 units of PCV were transfused and diuretic tablet (combination of Torsemide and Spiro lactone) one tablet daily was started. Next day of Caesarean Section left parotid gland swelling was observed with inflammation which was tackled with conservative treat ment.
Postpartum Day 3 when check dressing was done serosanguinous discharge was expressed out, whose cul ture and sensitivity was sent followed by daily antiseptic dressings.
Postpartum Day 8 wound gaping with giveaway of rec tus sheath was observed. Once the wound margins were healthy after proper cleaning and debridement of wound resuturing of wound using Prolene number 1 for rectus sheath and Vicryl number 1 for subcutaneous tissue and Sutu pack for skin closure was done. All sutures were taken as interrupted sutures. Stitch removal was done on 10th post-suturing day.
On Discharge - Iron, calcium, multivitamin tablets, pro tein powder along with Tab Azathioprine 50mg OD and Tab Prednisolone 10 mg OD were prescribed.
26 Years old Primigravida Known case of Cyanotic Con genital Heart Disease operated in 2005 at 10 years of age. Post Bidirectional Glenn (BDG) and Complete Fontan physiology with good functional status along with, Dextro cardia, Situs- Solitus, Double inlet Left Ventricle (DILV), Dominant left sided ventricle and Hypoplastic right ven tricle, Large Atrial Septal Defect (ASD), Large muscular and Subaortic Ventricular Septal Defect (VSD), well-func tioning Superior Vena Cava to Right Pulmonary Artery( SVC to RPA) Shunt, Severe Pulmonary Stenosis ,Single Atrioventricular valve with moderate left Atrioventricular (AV) Valve Regurgitation. She was maintaining Oxygen Saturation 97%. She was in remission of auto immune hepatitis when she came to us.
May 2016-She had high grade fever with loss of appetite and jaundice with high coloured urine. She was hospital ized and investigated. On investigations she detected to have Serum Bilirubin of 26.97mg/dL. INR was maintained to 1.3-1.44. Serum Proteins were reduced to 4.3 from 7.9 g/dL 8 g/dL. Various antibody tests for autoimmune disor ders were performed. The results were negative for ASMA, AMA, and ALKM, but positive for ANA.
| ANA | 1:100 POSITIVE |
| Pattern | ANA Pattern Nuclear Speckled Pattern |
| ASMA | Negative |
| AMA | Negative |
| ALKM | Negative |
| CMV125 | Positive |
| Ceruloplasmin | 50.23 Normal |
| HBc IgM | Negative |
| HSV I g M | Negative |
| EBV Viral Capsid Antigen IgM | Negative |
| HBsAg | Negative |
| Histo-Pathology | Cellular and canalicular cholestasis, lobular and moderate inflammation with lymphoplasma moderate interface activity and increased portal and periportal fibrosis suggestive of Auto immune Hepatitis |
Table 2: Laboratory and Diagnostic Findings for Case 2 Patient (Autoimmune Hepatitis with Cyanotic Congenital Heart Disease)

Figure 3: Transient elastography
Clinical Features- She had no ascites or flaps. She was on Tab Prednisolone 40 mg OD and her follow up was done every fortnightly for 3 months.
Total Serum Bilirubin levels decreased gradually over 4 months to normal range.
After one month on steroids, she developed right sided consolidated pneumonia which got resolved after giving higher antibiotics. Prednisolone was reduced to 30mg OD and Azathioprine 75 mg OD was added. She had a spontaneous conception. Antenatal period was uneventful with no relapse of AIH. During her pregnancy, liver function tests remained normal. She delivered a live female baby weighing 2.75 kg at 37 weeks 4 days gestation vaginally. Baby cried immediately after birth and postpartum period of the patient was uneventful.
Discussion
The natural history of AIH may progress from acute hepa titis to chronic liver disease and, ultimately, to cirrhosis. The probable theory for the pathogenesis of autoimmune hepatitis in pregnant women and the general population is that the disease results from an environmental trigger in a genetically predisposed individual. The precise mechan ism between the genes and the autoimmune process is still unknown. However, at the molecular level, they are believed to involve an antigen, a major histocompatibil ity complex (MHC), and a T cell receptor, which together form a complex in which complementary-determining re gions identify and contact the antigen-MHC complex. [11] The suggested environmental triggers that lead to this T cell-mediated immune response targeting the liver include viruses, medications, herbal supplements, and even im munization [12] or sometimes unidentifiable factor. The probable mechanism of obstetric complications in women with AIH is the targeted involvement of maternal-foetal interface structures by autoantibodies and natural immune cells activated by the disease. [13] These interface struc tures that get targeted include cellular components of inter villous space, endothelial tissue of spiral veins, and super ficial epithelial cells of the decidua. This process leads to an increased risk of first-trimester miscarriage and preterm delivery [13]. Thrombosis is another complication that oc curs due to endothelial damage and complement systemactivation, hence worsening of perinatal outcomes.
The clinical presentation of autoimmune hepatitis can be highly variable. It can present as either an acute or chronicillness with a fluctuating pattern. It can also be discovered incidentally in completely asymptomatic individuals on routine laboratory testing [14]. Patients can present with debilitating symptoms such as malaise, anorexia, fatigue, and weight loss. The patients can also present with acute liver failure as an initial presentation with jaundice, ascites, and coagulopathy [15] or normal physical examination. Autoimmune-mediated coexisting extrahepatic disorder may also be seen e.g. autoimmune thyroiditis, rheumatoid arthritis, systemic lupus erythematosus, type 1 diabetes mellitus, celiac disease, and ulcerative colitis [16].
Histo- Pathological Examination- Shows interface hepatitis, lymphocytic infiltration of the portal tracts ex tending into the lobule, emperipolesis, or active penetra tion of one cell into and through a large cell and hepatic rosette formation. The course of autoimmune hepatitis during pregnancy is unpredictable. Though the literature is limited, up to 20% of patients with AIH will flare during pregnancy, which is associated with a high rate of fetal and maternal complications [3]. Thus, women with AIH should be counselled to delay pregnancy at least one year until their disease is under optimal control [3]. Currently, pregnancy and childbirth in AIH appear to be safe for both mother and child. AIH may flare with the return of normal immunity after delivery, and, in fact, flares are twice as frequent in the postpartum period during pregnancy [3]. During flares, the management of pregnant patients with AIH should be similar to non-pregnant patients, with higher doses of prednisone and/or the addition of azathioprine. Cirrhosis can occur as an initial presentation at the time of AIH diagnosis in up to 40% of patients. Pregnancy in compensated cirrhotic women is no longer feared, nor is it contraindicated, but it correlates with increased preeclampsia rates, low birth weight, preterm delivery, and neonatal death with increased risk of maternal mortality and complications [17, 18]. Patients with a MELD (Model for End-Stage Liver Disease) score of greater than 10 are at higher risk for poor maternal and foetal outcomes, with an increased risk of decompensation and maternal mortality of up to 7.8% [18]. MELD score is based on 4 parameters i.e.INR (International Normalized Ratio), Creatinine, Bilirubin and Serum sodium levels [19]. This group of patients should have thorough discussions with their ob stetrician and hepatologist regarding elective pregnancy termination, surrogate pregnancy, and delaying pregnancy until after liver transplantation [20].
The reported incidence of flares is around 20% to 50% of patients postpartum. Therefore, recommendations are that the patient should be followed by a hepatologist closely. The patient’s liver function tests and immunoglobulin G (IgG) level should be monitored at delivery and then at 4 to 6-week intervals during the first three months postpartum [3]. Breastfeeding is safe with prednisone. Azathioprine is excreted at low levels in breast milk, and most experts recommend continuing azathioprine use during breastfeeding despite manufacturers’ recommendations against using them during lactation as advantages of breast feeding are more rather than disadvantages [10].
During pregnancy, compensatory dilation of collateral veins that join the portal circulation and the azygos vein occurs, causing expansion of the circulating blood volume and ex trinsic compression of the inferior vena cava by the preg nant uterus, leading to increased portal hypertension and variceal bleeding, especially during the third trimester and labour and delivery [20]. This situation is the most common complication and cause of maternal morbidity and mortal ity. Thus, all patients should have an upper endoscopy to identify and potential prophylactic endoscopic variceal li gation of high-risk varices – especially around 28 weeks of gestation when maximum volume expansion occurs [3].
Autoimmune hepatitis has an array of clinical phenotypes. For this reason, it appears in the differential diagnosis for patients presenting with abnormal liver biochemical tests, cirrhosis, acute hepatitis, or acute liver failure [21]. Furthermore, even with the establishment of diagnostic criteria, there are still issues to be clarified attributable to its variability, such as autoantibody-negative AIH, drug-in duced AIH, AIH sharing features with primary biliary cir rhosis (PBC) or primary sclerosing cholangitis (PSC) (i.e., Overlap Syndrome), and also rarely, post-transplant de novo AIH.
These patients come under vulnerable high-risk group. Risk of surgical site or variceal bleed, jaundice, ascites or hepatic encephalopathy can manifest in them along with high risk of acquiring opportunistic infections as they are on immunosuppressants and their serum proteins are on lower side along with anaemia of chronic disease, so special antenatal care along with perinatal surveillance are necessary for good outcome of pregnancy.
Conclusion
Such high-risk cases should be tackled by a multidisciplin ary approach involving Obstetrician, Hepatologist, Cardi-ologist, Anaesthesiologist and Neonatologist taking into account all possible complications like hepatitis relapse, thrombotic events, inflammatory disorders and activation of autoimmune system. So, it is very important to counsel patients carefully on the potential complications before conception so that patient is mentally prepared for regu lar follow up with clinician. Hence close surveillance along with clinical, medical and technological improvements are associated with positive outcomes for both mother and baby.
References