Moses Adondua Abah1,2*, Esther Edesiri Ajoku3, Odusanya Kikunlore Elijah4, Micheal Abimbola Oladosu2,5,Ekpenyong Utomobong Sunday6, Ejim Thomas Ejim7 and Nathan Rimamsanati Yohanna1,2
1Department of Biochemistry, Faculty of Biosciences,Federal University Wukari, Wukari, Taraba State,Nigeria
2ResearchHub Nexus Institute, Nigeria
3Healthcare Management and Administration, University of New Orleans, 2000 Lakeshore Drive, New Orleans, la 70148, United States of America
4 Department of Hematology and Blood Transfusion, Olabisi Onabanjo University Teaching Hospital, Sagamu, Ogun State, Nigeria
5Department of Biochemistry, Faculty of Basic Medical Sciences, University of Lagos, Idi-Araba, Nigeria
6Department of Internal Medicine, College of Medicine, University of Uvo Teaching Hospital, Akwa-Ibom State, Nigeria
7 Department of Surgery, University College Hospital, Ibadan, Oyo State, Nigeria
*Correspondence: Moses Adondua Abah, Department of Biochemistry, Faculty of Biosciences, Federal University Wukari, Research Hub Nexus Institute, Nigeria E-mail: m.abah@fuwukari.edu.ng
Received: 21 Oct, 2025; Accepted: 12 Nov, 2025; Published: 27 Nov, 2025
Citation: Moses Adondua Abah, Esther Edesiri Ajoku, Ekele Angel Ojimaojo, Ekpenyong Utomobong Sunday, Captain Tamunotonye Blessing, Michael Abimbola Oladosu, John Oladipo Olutayo and Nkwopara peace Chisa. “Non-Alcoholic Fatty Liver Disease (NAFLD) and Metabolic Syndrome: An Intertwined Epidemic” J Pathol Diagn Microbiol(2025). DOI:10.59462/JPDM.1.1.102
Copyright: © 2025 Moses Adondua Abah. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Non-Alcoholic Fatty Liver Disease (NAFLD) and Metabolic Syndrome (MetS) are two interconnected health conditions that have become increasingly prevalent worldwide. NAFLD is characterized by excessive fat accumulation in the liver, while MetS is a cluster of conditions including obesity, insulin resistance, hypertension, and dyslipidemia. The two conditions share common pathophysiological mechanisms and risk factors, and individuals with NAFLD are more likely to develop MetS and vice versa. The growing burden of NAFLD and MetS poses significant challenges to global health, highlighting the need for a comprehensive understanding of their relationship and effective management strategies. This review aimed at providing a comprehensive overview of the complex relationship between NAFLD and MetS, highlighting the shared pathophysiological mechanisms, diagnostic challenges, and therapeutic implications. Findings from this study reveals a strong bidirectional relationship between Non-Alcoholic Fatty Liver Disease (NAFLD) and Metabolic Syndrome (MetS). NAFLD is recognized as the hepatic manifestation of MetS, with insulin resistance being a key pathophysiological link. Studies show that NAFLD prevalence is higher in individuals with MetS, and vice versa. The presence of NAFLD exacerbates MetS components, including obesity, dyslipidemia, and hypertension. Conversely, MetS worsens NAFLD progression, increasing the risk of advanced fibrosis and cirrhosis. The intertwined nature of NAFLD and MetS necessitates a comprehensive approach to diagnosis, treatment, and prevention. The intertwined epidemic of NAFLD and MetS demands a holistic approach to management, incorporating lifestyle modifications, pharmacological interventions, and targeted therapies. Early diagnosis and treatment of both conditions can mitigate disease progression, reducing the risk of cardiovascular and liver-related complications. A multidisciplinary approach, involving hepatologists, endocrinologists, and primary care physicians, is essential to address this complex health issue and improve patient outcomes.
Keywords:
Non-Alcoholic Fatty Liver Disease (NAFLD), Metabolic Syndrome (MetS), Insulin, Obesity, Liver, and Cardiovascular disease
Introduction
Non-Alcoholic Fatty Liver Disease (NAFLD) and Metabolic Syndrome (MetS) have surfaced as two of the most significant public health issues of the 21st century, closely connected through common pathophysiological processes and lifestyle factors. NAFLD is characterized by the buildup of excess fat in the liver that is not linked to alcohol intake or other secondary causes of hepatic steatosis [1]. It includes a range of conditions from simple steatosis to non-alcoholic steatohepatitis (NASH), fibrosis, and potentially cirrhosis or hepatocellular carcinoma. Conversely, Metabolic Syndrome is defined as a combination of metabolic disorders such as central obesity, insulin resistance, dyslipidemia, and hypertension, which together elevate the risk of cardiovascular disease and type 2 diabetes [2]. These conditions are not only biologically intertwined but also epidemiologically convergent, with rising prevalence driven by urbanization, sedentary lifestyles, and dietary transitions [3]. Globally, the burden of NAFLD has surged dramatically, with recent estimates indicating that over 1.2 billion individuals were affected in 2021, marking a 124.6% increase since 1990. Age-standardized prevalence rates rose from 12,085 to 15,018 per 100,000, with the highest incidence observed in Oceania, Central and Latin America, and the Middle East [4]. In Nigeria, the picture is equally concerning. A study conducted at the University College Hospital in Ibadan found that 46% of patients with type 2 diabetes had NAFLD, highlighting a significant overlap between hepatic steatosis and metabolic dysfunction in the Nigerian population [5]. Similarly, the prevalence of MetS in Nigeria has escalated in recent decades, reflecting the country’s rapid urbanization and dietary shifts. These figures underscore the urgent need for integrated strategies that address both liver health and metabolic regulation.
Understanding the relationship between NAFLD and MetS is crucial because they often coexist and amplify each other’s clinical consequences. NAFLD is increasingly recognized as the hepatic manifestation of MetS, and its presence can accelerate insulin resistance, exacerbate lipid abnormalities, and heighten cardiovascular risk [2]. Conversely, the components of MetS particularly central obesity and hyperglycemia are potent drivers of hepatic fat accumulation and inflammation. This bidirectional interplay complicates diagnosis, treatment, and prognosis, making it imperative for clinicians and researchers to approach these conditions as a unified epidemic rather than isolated entities.
The increasing awareness of the prevalence and synergistic impact of NAFLD and MetS, especially in low- and middleincome nations like Nigeria where healthcare systems are underfunded, is what spurred this research [2]. In order to inform focused initiatives and policy frameworks, this study will synthesize current epidemiological data, investigate common causes, and identify clinical management gaps. Fostering a more comprehensive understanding of this interconnected disease and accelerating efforts towards lifestyle changes, early identification, and integrated treatment solutions that can lessen its long-term effects are the objectives.
NAFLD begins with the excessive accumulation of triglycerides within hepatocytes, a process largely driven by insulin resistance. Under normal physiological conditions, insulin suppresses lipolysis in adipose tissue and promotes glucose uptake and lipid storage. However, in insulinresistant states, there is an unchecked release of free fatty acids (FFAs) from adipose tissue into the circulation [6]. These FFAs are taken up by the liver, where they are either re-esterified into triglycerides or oxidized. When the influx of FFAs exceeds the liver’s oxidative capacity, triglycerides accumulate, leading to hepatic steatosis [7]. Figure 1 vividly illustrates this cascade, showing how insulin resistance disrupts the hormonal balance between the pancreas, adipose tissue, and liver.
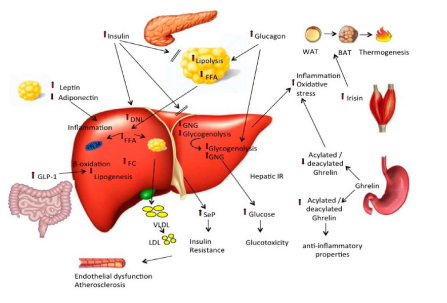
Figure 1: Pathophysiology of Non-Alcoholic Fatty Liver Disease (NAFLD)
The pancreas, despite secreting insulin, fails to suppress lipolysis in adipose tissue, resulting in elevated FFAs that flood the liver. Simultaneously, hepatic de novo lipogenesis is upregulated due to impaired insulin signaling, compounding the lipid burden. As depicted, the liver responds to this overload with increased lipogenesis and reduced β-oxidation, setting the stage for lipotoxicity. This toxic lipid environment triggers mitochondrial dysfunction and oxidative stress, which are central to hepatocellular injury [3]. The diagram also highlights the role of inflammatory mediators originating from both adipose tissue and the liver itself that amplify this damage. Adipokines such as leptin and adiponectin, along with gutderived hormones like GLP-1 and ghrelin, modulate this inflammatory response. In particular, the acylated form of ghrelin and reduced adiponectin levels contribute to a proinflammatory milieu, while GLP-1 offers a counterbalance with its insulin-sensitizing and anti-inflammatory effects [2]. Moreover, the figure underscores the systemic nature of NAFLD, linking hepatic lipid metabolism to cardiovascular risk via the production of VLDL and LDL particles [3]. These lipoproteins, exported from the liver, contribute to atherosclerosis and endothelial dysfunction, reinforcing the notion that NAFLD is not confined to the liver but reflects a broader metabolic derangement. Ultimately, the interplay of insulin resistance, disrupted lipid metabolism, and chronic inflammation captured in the dynamic flow of Figure 1 drives the progression from simple steatosis to non-alcoholic steatohepatitis (NASH) and beyond [3].
Metabolic Syndrome shares insulin resistance as its central pathophysiological feature, but its manifestations extend beyond the liver to encompass systemic metabolic dysfunction. In MetS, insulin resistance contributes to hyperglycemia, dyslipidemia, and hypertension [3]. Adipose tissue, particularly visceral fat, becomes metabolically active, secreting adipokines and inflammatory mediators that exacerbate systemic insulin resistance. Leptin resistance and reduced adiponectin levels impair glucose and lipid metabolism, while elevated levels of TNF-α and IL-6 promote chronic low-grade inflammation [8]. This inflammatory state not only worsens insulin resistance but also contributes to endothelial dysfunction, a key driver of cardiovascular disease in MetS.
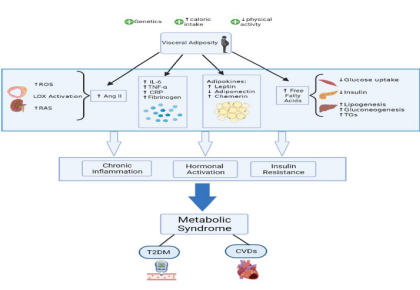
Figure 2:The central role of insulin resistance, visceral adiposity, and chronic inflammation in the development of MetS
Figure 2 visually reinforces the central role of obesity particularly visceral fat as the initiating factor in MetS. As adipose tissue expands, adipocytes undergo hypertrophy and experience hypoxia, triggering apoptosis and attracting macrophages that polarize toward a pro-inflammatory M1 phenotype. These immune cells release cytokines like TNF-α and IL-6, which impair insulin signaling and fuel systemic inflammation (1). The diagram shows how this inflammatory milieu is compounded by increased reactive oxygen species (ROS), activation of lipoxygenase (LOX), and the renin-angiotensin system (RAS), leading to elevated Angiotensin II and vascular dysfunction. Simultaneously, the metabolically active adipose tissue alters adipokine profiles raising leptin, lowering adiponectin, and increasing chemerin disrupting glucose and lipid homeostasis [3]. Free fatty acids (FFAs) released from adipose stores spill into circulation, contributing to ectopic fat deposition in the liver, pancreas, and muscle, which further exacerbates insulin resistance. The image captures how these changes culminate in decreased glucose uptake and increased hepatic lipogenesis and gluconeogenesis, driving hyperglycemia and dyslipidemia. Ultimately, the convergence of chronic inflammation, hormonal imbalance, and insulin resistance leads to the full clinical expression of MetS, with heightened risk for type 2 diabetes mellitus and cardiovascular diseases. Figure 2 encapsulates this systemic dysfunction, offering a clear and integrated view of how central obesity orchestrates the metabolic collapse characteristic of MetS [10].
The shared mechanisms between NAFLD and MetS are rooted in common genetic predispositions and environmental exposures. Polymorphisms in genes such as Patatin-like phospholipase domain-containing protein 3 (PNPLA3), Transmembrane 6 superfamily member 2 (TM6SF2), and Membrane-bound O-acyltransferase domain-containing 7 (MBOAT7) have been implicated in both NAFLD susceptibility and metabolic traits, highlighting a genetic overlap [10,11]. For instance, the PNPLA3 I148M variant is strongly associated with hepatic fat accumulation and progression to NASH, independent of obesity and insulin resistance. Environmental factors, including highcalorie diets rich in saturated fats and refined sugars, sedentary behavior, and urbanization, further amplify these genetic risks. The Western dietary pattern promotes gut dysbiosis and increased intestinal permeability, allowing endotoxins such as lipopolysaccharide (LPS) to enter the portal circulation and activate hepatic Toll-like receptors, thereby fueling inflammation and insulin resistance [2].
Epidemiological data emphasize how strong the link is between non-alcoholic fatty liver disease (NAFLD) and metabolic syndrome (MetS). Globally, NAFLD affects approximately 25% of the general population, but its prevalence surges to over 50% among individuals diagnosed with MetS [10]. Conversely, MetS is frequently observed in patients with NAFLD, with rates often exceeding 40%, depending on demographic factors and diagnostic criteria. This bidirectional relationship is particularly pronounced in regions undergoing rapid urbanization and lifestyle transitions, such as sub-Saharan Africa. For instance, hospital-based research in Nigeria revealed that nearly half of patients with type 2 diabetes also present with NAFLD, underscoring the metabolichepatic overlap [5]. Table 1 provides a comparative summary of prevalence rates across various populations, highlighting the mutual amplification of risk. It illustrates that individuals with MetS are significantly more likely to develop NAFLD, and vice versa, reinforcing the concept of a shared pathophysiological basis. The table also includes regional data, such as findings from Nigerian cohorts, which reflect the growing burden of these conditions in developing nations. This epidemiological convergence is rooted in common mechanisms namely insulin resistance, chronic low-grade inflammation, and adipose tissue dysfunction suggesting that integrated strategies targeting these pathways may be essential for effective prevention and management [10]
|
Condition |
Associated Prevalence |
Prevalence |
|
NAFLD in individuals with MetS |
50–90% |
NAFLD is highly prevalent among those with MetS due to shared risk factors. |
|
MetS in individuals with NAFLD |
30–60% |
Many NAFLD patients meet MetS criteria, especially with central obesity. |
|
Shared Risk Factors |
Obesity, insulin resistance, dyslipidemia, hypertension |
These drive both conditions and explain their bidirectional relationship. |
|
Strongest Predictors |
Visceral fat, elevated triglycerides, impaired glucose |
These are common markers in both NAFLD and MetS. |
|
Clinical Implication |
NAFLD may be considered the hepatic manifestation of MetS |
Early detection of one condition can prompt screening for the other. |
Table 1:Bidirectional prevalence of NAFLD and metabolic syndrome across populations
There is growing evidence from mechanistic insights that the link between NAFLD and MetS is bidirectional. Through hepatic insulin resistance, which impairs glucose and lipid metabolism, NAFLD plays a role in the development of MetS. Triglyceride buildup in hepatocytes disrupts insulin-signaling pathways, resulting in hyperglycemia and dyslipidemia, two major symptoms of metabolic syndrome [12]. Furthermore, NAFLD is linked to elevated release of pro-inflammatory cytokines such interleukin-6 (IL-6) and tumour necrosis factor-alpha (TNF-α), which worsen insulin resistance and systemic inflammation. However, because of adipose tissue malfunction, MetS increases the amount of free fatty acids that enter the liver, hastening the onset of non-alcoholic fatty liver disease.
Adipocyte hypertrophy and hypoxia brought on by central obesity, a defining feature of MetS, cause macrophage infiltration and a change in phenotype towards one that is pro-inflammatory. These immunological reactions worsen hepatic insulin sensitivity and encourage the growth of fibrosis and steatohepatitis [1,13]. A common basis for the co-occurrence of NAFLD and MetS is provided by their shared risk factors. The most powerful cause of these disorders is obesity, especially visceral adiposity. Overweight causes metabolic dysregulation by upsetting adipokine balance, lowering adiponectin levels, and raising leptin resistance [12]. By decreasing insulin sensitivity and encouraging ectopic fat deposition, physical inactivity exacerbates these consequences. It has been demonstrated that dietary patterns high in fructose, refined carbs, and saturated fats exacerbate metabolic profiles and hasten the buildup of hepatic fat. The pathophysiology of both NAFLD and MetS has been linked to the Western diet, which is marked by a high calorie density and low fiber intake. Similar to worldwide patterns, Nigeria has seen a shift in diet towards processed foods and a decline in physical activity in urban areas, which has led to an increase in the prevalence of both diseases [14].
The screening and diagnosis of NAFLD and MetS represent one of the most significant clinical difficulties. Early-stage NAFLD frequently exhibits no symptoms, and the diagnosis of the disease is usually made using imaging techniques like ultrasound, which, although readily available, is insensitive in identifying mild steatosis or distinguishing between simple fatty liver and more severe types like non-alcoholic steatohepatitis (NASH). Although liver biopsy is still the gold standard for conclusive diagnosis, its widespread use is limited by its invasiveness, expense, and related dangers. Although transient elastography and serum biomarkers are promising non-invasive technologies, their availability and standardization in various healthcare settings are still uneven [12]. Contrarily, MetS is diagnosed using clinical criteria such blood pressure, fasting glucose, triglycerides, waist circumference, and HDL cholesterol levels. However, these parameters can differ between populations and guidelines, which could result in an underdiagnoses or incorrect classification. Early identification is made more difficult in Nigeria by a lack of access to thorough metabolic screening and inequalities in the country’s healthcare system, particularly in underprivileged and rural areas.
NAFLD and MetS have significant and varied clinical repercussions. Among those with NAFLD, cardiovascular disease (CVD) continues to be the primary cause of death, outpacing liver-related deaths. Hepatic steatosis produces an environment that is both pro-inflammatory and pro atherogenic, which leads to endothelial dysfunction, arterial stiffness, and accelerated atherosclerosis. Increased carotid intima-media thickness and coronary artery calcification, which are indicators of subclinical CVD and frequently occur before overt events like myocardial infarction and stroke, are seen in patients with NAFLD [12]. Because insulin resistance, hypertension, and dyslipidemia all work together to increase vascular damage and thrombosis, MetS exacerbates this risk. Another significant effect of NAFLD is type 2 diabetes mellitus (T2DM), which is both a cause and a predictor of impaired glucose metabolism. The onset of type 2 diabetes is accelerated by hepatic insulin resistance, which upsets glucose homeostasis, and chronic inflammation and lipotoxicity, which affect pancreatic beta-cell function. The advancement of NAFLD, including cirrhosis and fibrosis, is more likely to occur in those with type 2 diabetes, on the other [15].
The most obvious hepatic effect of untreated NAFLD is advanced liver disease. Even while mild steatosis can last for years, some patients develop NASH, which is marked by inflammation and liver damage. It is possible for NASH to progress to cirrhosis, fibrosis, and finally hepatocellular carcinoma (HCC) without substantial alcohol consumption [12]. The fact that this trend is quiet emphasizes how crucial risk classification and attentive monitoring are. The increasing prevalence of NAFLD-related cirrhosis may go unnoticed in Nigeria, where viral hepatitis is frequently blamed for liver disease. This could result in missed opportunities for intervention and delays in diagnosis.
It is impossible to overestimate the significance of early detection and treatment in light of these clinical implications. When at-risk individuals are identified early, lifestyle measures that have been demonstrated to reverse hepatic steatosis and improve metabolic parameters, such as weight loss, dietary adjustments, and increased physical activity, can be implemented. Although no particular medication is currently approved for the treatment of NAFLD, pharmacological therapies such as insulin sensitizers and lipid-lowering medications may be taken into consideration in some situations [12]. Comprehensive management requires a multidisciplinary, holistic approach that incorporates primary care, cardiology, endocrinology, and hepatology. In order to enable people and communities to take preventative action, public health initiatives must also place a high priority on education, screening, and awareness.
For both NAFLD and MetS, lifestyle change continues to be the mainstay of care [16,17]. Losing weight has been demonstrated to improve hepatic steatosis, lower insulin resistance, and improve cardiovascular risk factors, making it one of the most effective therapies. Long-term weight loss of 7–10% is linked to notable histological improvements in liver fibrosis and inflammation [17]. Changes in diet are essential to reaching this objective. According to [16], the Mediterranean diet, which is high in monounsaturated fats, whole grains, fruits, vegetables, and lean proteins, has been shown to be effective in lowering liver fat and enhancing metabolic parameters.
On the other hand, diets heavy in processed foods’ fructose, refined carbs, and saturated fats worsen insulin resistance and hepatic lipid buildup [17]. Dietary education and access to nutritious foods are public health priorities in Nigeria, as the country’s shift to energy-dense, lowfiber diets has increased the prevalence of NAFLD and MetS [17]. By increasing insulin sensitivity, encouraging fat oxidation, and decreasing visceral adiposity, physical exercise enhances dietary treatments [16]. Independent of weight loss, aerobic exercise—such as brisk walking, cycling, or swimming has been linked to decreases in liver fat when done for at least 150 minutes each week [17].
Additionally, resistance exercise enhances muscle growth and glycemic management, both of which promote metabolic health. Significantly, physical exercise lowers the risk of cardiovascular disease and type 2 diabetes, which has advantages beyond liver results [16]. Promoting accessible and culturally relevant types of exercise, including dance-based fitness or community walking groups, can improve sustainability and adherence in environments with low resources [17]. Although lifestyle modifications are fundamental, they are frequently insufficient for people with severe metabolic disorders or advanced disease. Additional therapy alternatives are provided by pharmacological interventions, especially for patients who have concomitant conditions like dyslipidaemia or type 2 diabetes.
Although worries regarding weight gain and long-term safety still exist, insulin sensitisers, such as pioglitazone, have demonstrated promise in improving liver histology in individuals with non-alcoholic steatohepatitis (NASH) [19]. Liraglutide and semaglutide are examples of glucagonlike peptide-1 receptor agonist (GLP-1 RAs), which are appealing choices for patients with NAFLD and MetS since they not only enhance glycaemic control but also encourage weight loss and liver fat reduction. In patients with diabetes, sodium-glucose cotransporter-2 (SGLT2) inhibitors have also been shown to help lower cardiovascular risk and hepatic steatosis [18]. Although worries regarding weight gain and long-term safety still exist, insulin sensitisers, such as pioglitazone, have demonstrated promise in improving liver histology in individuals with non-alcoholic steatohepatitis (NASH). Liraglutide and semaglutide are examples of glucagon-like peptide-1 receptor agonist (GLP-1 RAs), which are appealing choices for patients with NAFLD and MetS since they not only enhance glycemic control but also encourage weight loss and liver fat reduction. In patients with diabetes, sodium-glucose cotransporter-2 (SGLT2) inhibitors have also been shown to help lower cardiovascular risk and hepatic steatosis [18].
Treatment for NAFLD and MetS is changing as a result of new therapeutic approaches. Currently being researched are novel pharmacological drugs that target lipid metabolism, fibrosis, and inflammation [19]. Obeticholic acid and other farnesoid X receptor (FXR) agonists alter bile acid signaling and have demonstrated potential in lowering fibrosis in NASH patients. The metabolic and hepatic benefits of other substances, such as fibroblast growth factor analogues and beta agonists of the thyroid hormone receptor, are being investigated [20]. Many of these treatments are still in the clinical trial stage, and their long-term safety and effectiveness have not yet been completely determined, despite promising results. For individuals with severe obesity and resistant metabolic disorders, bariatric surgery is a potent strategy. Sleeve gastrectomy and Roux-en-Y gastric bypass are two procedures that frequently result in significant and longlasting weight loss, improved insulin sensitivity, and the treatment of NAFLD [21,22]. Research has demonstrated that bariatric surgery can reverse cirrhosis and even fibrosis after the procedure, suggesting that it may be a treatment option for severe liver disease [22]. However, in many lowand middle-income nations, including Nigeria, access to surgical care is still restricted, and successful outcomes depend on careful patient selection and postoperative care [23].
The future of managing Non-Alcoholic Fatty Liver Disease (NAFLD) and Metabolic Syndrome (MetS) hinges on closing critical research gaps, identifying novel therapeutic targets, and translating scientific advances into meaningful clinical and public health strategies [12]. As these conditions continue to rise in prevalence particularly in low- and middle-income countries like Nigeria the urgency to deepen our understanding and refine our interventions becomes paramount.
Despite significant progress, several research gaps persist in the pathophysiology of NAFLD and MetS. The precise molecular mechanisms driving the transition from simple hepatic steatosis to non-alcoholic steatohepatitis (NASH), fibrosis, and hepatocellular carcinoma remain incompletely understood [12]. While insulin resistance and chronic inflammation are recognized as central contributors, the interplay between genetic predisposition, epigenetic modifications, and environmental factors warrants further exploration. Additionally, the heterogeneity of NAFLD presentations ranging from metabolically healthy steatosis to aggressive fibrotic disease suggests the need for more nuanced disease classification systems. Current diagnostic tools, including imaging and liver biopsy, are limited in their ability to predict disease progression or response to therapy. The development of non-invasive, reliable biomarkers for early detection and risk stratification remains a critical unmet need [24].
Emerging research is beginning to uncover promising therapeutic targets that may revolutionize treatment. Molecular pathways involved in lipid metabolism, inflammation, and fibrosis are being actively investigated. Farnesoid X receptor (FXR) agonists, such as obeticholic acid, modulate bile acid signaling and have shown potential in reducing liver fibrosis [12]. Peroxisome proliferatoractivated receptor (PPAR) agonists, including elafibranor, target lipid synthesis and inflammation with encouraging results in clinical trials. Other agents, such as fibroblast growth factor analogs and acetyl-CoA carboxylase inhibitors, aim to reduce hepatic lipogenesis and improve insulin sensitivity [17]. Biomarkers such as cytokeratin-18 fragments, microRNAs, and metabolomic profiles are being explored for their diagnostic and prognostic utility. The integration of omics technologies genomics, proteomics, and metabolomics offers a pathway toward precision medicine, enabling tailored interventions based on individual risk profiles [19].
The implications of these advances for clinical practice are profound. A shift toward personalized care models, informed by molecular diagnostics and targeted therapies, could enhance treatment efficacy and reduce adverse outcomes [12]. Clinicians will need to adopt multidisciplinary approaches that incorporate hepatology, endocrinology, cardiology, and nutrition to manage the complex interplay of metabolic and hepatic dysfunction [25]. In Nigeria and similar settings, strengthening diagnostic infrastructure and training healthcare providers in emerging technologies will be essential to bridge the gap between research and practice [12].
From a public health perspective, the growing burden of NAFLD and MetS demands policy-level interventions. Governments must prioritize funding for research, support the development of national screening programs, and implement population-wide strategies to promote healthy lifestyles [25]. Public education campaigns focused on nutrition, physical activity, and early detection can help curb the epidemic. Additionally, policies that regulate food labeling, urban planning for physical activity, and access to affordable healthcare will be instrumental in addressing the social determinants of metabolic disease [12].
The interconnectedness of Metabolic Syndrome (MetS) and Non-Alcoholic Fatty Liver Disease (NAFLD) is a reflection of the convergence of lifestyle-driven, inflammatory, and metabolic processes that have changed the burden of chronic disease worldwide. The common pathophysiological processes that underlie both disorders have been emphasised in this study, including insulin resistance, visceral obesity, and chronic inflammation. It has also highlighted the reciprocal interaction between NAFLD and MetS, in which both conditions actively contribute to the severity and advancement of the other in addition to coexisting. The severe clinical repercussions, which include advanced liver pathology, cardiovascular disease, and type 2 diabetes, highlight the necessity of combined diagnostic and treatment approaches.
It is crucial for researchers, physicians, and policymakers to comprehend the connection between NAFLD and MetS. It is crucial to treat them as parts of a single metabolic disorder rather than as separate conditions since their co-occurrence increases the burden of the disease and makes management more difficult. In order to address both hepatic and systemic problems, this viewpoint promotes interdisciplinary care, early screening, and tailored interventions. Future studies must concentrate on determining the molecular heterogeneity of MetS and NAFLD, finding trustworthy non-invasive biomarkers, and creating focused treatments that deal with metabolic imbalance, fibrosis, and inflammation. Prevention should be the top priority of public health initiatives through lifestyle promotion, education, and fair access to care. It would be crucial to incorporate metabolic illness treatment into primary care and community health initiatives in areas like Nigeria, where the combined burden of infectious and noncommunicable diseases puts a pressure on healthcare systems. We may respond to this interconnected epidemic in a more efficient and inclusive manner by combining scientific understanding with clinical practice and legislative change.
To distill the key findings, the following (Table 2) summarizes the major insights from this review:
|
Key Domain |
Summary of Findings |
|
Pathophysiology |
Insulin resistance, lipid dysregulation, and inflammation drive both NAFLD and MetS. |
|
Epidemiology |
NAFLD affects ~25% globally; prevalence exceeds 50% among individuals with MetS. |
|
Bidirectional Relationship |
NAFLD promotes insulin resistance and inflammation; MetS accelerates hepatic fat buildup. |
|
Clinical Implications |
Increased risk of cardiovascular disease, type 2 diabetes, and liver fibrosis/cirrhosis. |
|
Management Strategies |
Lifestyle modification is foundational; pharmacologic and surgical options are evolving. |
|
Research and Policy Gaps |
Need for biomarkers, targeted therapies, and public health infrastructure. |
Table 2: Key findings
We thank all the researchers who contributed to the success of this research work.
The authors declared that there are no conflicts of interest.
No funding was received for this research work