Joshua Sungho Hong 1,2*,Radha Nair1
1Swan Hill District Health, Swan Hill, Victoria, Australia
2St. Vincent’s Hospital Melbourne, Victoria, Australia
Received: 223 May 2025; Accepted: 16 June 2025; Published: 23 June 2025
Citation: Joshua Sungho Hong,Nair R. “Strategies to Adopt in a Challenging Laparoscopic Cholecystectomy” J Aesthetic Surg Med (2025): 108. DOI: 10.59462/ JASM.2.1.108
Copyright: © 2025 Joshua Sungho Hong. This is an open-access arti cle distributed under the terms of the Creative Com mons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Laparoscopic Cholecystectomy [LC] is among the most performed surgical procedures worldwide. While typically straightforward, a subset of cases presents significant technical difficulty due to inflammation, fibrosis, or anatomical distortionfactors that dramatically increase the risk of bile duct injury [BDI], conversion to open surgery, and other complications. Risk factors of a challenging cholecystectomy are male sex, older age, obesity, acute or recurrent cholecystitis, cirrhosis, and anatomical anomalies. A strong emphasis is placed on hepatobiliary anatomy, including variants of the cystic duct, cystic artery, Rouviere’s sulcus, and accessory bile ducts, which play a critical role in surgical safety. Mirizzi’s syndrome, though rare, represents a particularly hazardous scenario and is discussed in detail with respect to its classification and operative implications. Core surgical strategies are reviewed, highlighting the Critical View of Safety [CVS], liberal use of intraoperative cholangiography, and the value of intraoperative pauses. Bailout techniques, including subtotal cholecystectomy, fundus-first dissection, and conversion to open surgery, are presented as essential tools to mitigate risk when standard dissection is unsafe or unachievable. Subtotal cholecystectomy is recognised as a key damagecontrol option with excellent outcomes in expert hands. Emerging adjuncts such as Indocyanine Green [ICG] fluorescence cholangiography, robotic surgery, and early artificial intelligence technologies are explored for their roles in enhancing intraoperative visualisation and decision-making.
Ultimately, safe management of the difficult gallbladder requires anatomical expertise, a culture of intraoperative vigilance, and readiness to adapt when standard approaches are inadequate. This review aims to equip surgeons with a structured framework to anticipate difficulty, avoid injury, and optimise outcomes in complex cholecystectomy scenarios.
Laparoscopic Cholecystectomy; gallbladder; Mirizzi’s syndrome; subtotal cholecystectomy; Anatomy
Laparoscopic Cholecystectomy [LC] is one of the most common operations performed worldwide, with hundreds of thousands done annually [1]. Laparoscopic Cholecystectomy [LC] has become the gold-standard for treating symptomatic gallstone disease, resulting in reduced post-operative pain, orbidity, and faster recovery compared to open surgery [2]. Despite the routine nature of LC, a subset of cases are deemed “difficult” or challenging, posing significant clinical importance [3]. A difficult LC is associated with longer operative times, higher conversion to open surgery, increased complication rates [including bile duct injury], and extended hospital stay. Difficult LC are generally classified as the ones with severe inflammation or scarring distorts the biliary anatomy and complicates dissection.
Situations like acute calculous cholecystitis [especially if gangrenous or emphysematous], gallbladder perforation, Mirizzi syndrome [an impacted stone causing biliary compression or fistula], or chronic fibrosis can obscure Calot’s triangle and make identification of structures challenging [4]. In one large review [>300,000 patients], factors like acute or chronic cholecystitis, “bad gallbladder” pathology [necrosis, gangrene, etc.], cystic duct stones, anatomical variants, and cholecystoenteric fistulas were all linked to difficult cholecystectomy [5]. Risk factors associated with a difficult LC includes male sex, age, acute on chronic cholecystitis, obesity and cirrhosis of the liver [6-8]. LC is the leading cause of Bile Duct Injury [BDI] in surgery, and the vast majority of major BDIs occur in these difficult scenarios with severe inflammation or aberrant anatomy [9]. BDIs can have significant morbidity impacts on patients. Therefore, understanding how to predict, prepare for, and manage a challenging cholecystectomy is of a paramount clinical significance. This paper provides a comprehensive review of difficult cholecystectomy management-including pertinent anatomy, evidence-based guidelines, surgical techniques, bail-out strategies, and emerging technologies.
A thorough knowledge of the hepatobiliary anatomy and its many variations is critical for safely navigating a difficult cholecystectomy. Key anatomic relationships in the hepatocystic [Calot’s] triangle are paramount. The classic Calot’s triangle is bounded by the cystic duct, common hepatic duct, and inferior surface of the liver; it typically contains the cystic artery, lymph node of Lund, and fatty areolar tissue [10]. In reality, the upper boundary is the liver, and surgeons refer to the “Calot’s” or hepatocystic triangle as the space that must be cleared to achieve the critical view of safety [11]. Inflammation [acute or chronic] can fill this triangle with fibrous tissue and adhesions, obscuring the normal landmarks [12]. Scarring may adhere the gallbladder to surrounding structures, making the cystic duct and artery difficult to identify. Important anatomical structures and variants to consider include:
Proper identification of the cystic duct and cystic artery within Calot’s triangle is paramount. The “Critical View of Safety” [CVS] technique entails clearing all fat and fibrous tissue from Calot’s triangle, exposing the cystic duct and artery entering the gallbladder, and confirming the gallbladder is separated from the liver bed except at the cystic structures [13]. In difficult cases, achieving the CVS can be challenging but remains the gold standard for avoiding misidentification injuries. An inflamed lymph node of Lund or dense adhesions may be encountered and should be carefully dissected to reveal the cystic artery beneath [11].
Rouviere’s sulcus is a natural fissure on the liver’s undersurface which is present in ~80% of individuals that corresponds to the plane of the right porta hepatis [14]. It runs to the right of the hepatic hilum and, when visible, serves as a safety landmark during cholecystectomy. A general rule is to keep dissection above the level of Rouviere’s sulcus to avoid injuring the common bile duct which lies below that plane. Drawing an imaginary line from the sulcus to the umbilical fissure [the R4U line] demarcates a safe zone for dissection on the gallbladder versus the danger zone deeper toward the porta hepatis [12]. In a hostile anatomy scenario, identifying Rouviere’s sulcus can help orient the surgeon when usual landmarks are lost.
In about 70-80% of cases, the cystic artery arises from the right hepatic artery and traverses within Calot’s triangle [15]. Commonly it bifurcates into superficial and deep branches to the gallbladder. A notorious variation is a short cystic artery arising from a tortuous right hepatic artery that loops [sometimes termed a Moynihan’s hump or caterpillar turn]. The right hepatic artery may be mistaken for the cystic artery because it gives off a very short cystic branch. A catastrophic injury could arise if one erroneously clips the right hepatic artery [16]. Therefore, caution should be taken when the “cystic artery” seems unusually large or low in the field, as it could potentially be an aberrant or early-branching right hepatic artery [17].
An accessory cystic artery might arise from the gastroduodenal artery or left hepatic artery. The cystic artery usually passes behind the cystic duct [retroductal], but it can run anterior to the duct in around 4–5% of cases [18].
There are numerous biliary tree anomalies that can make a cholecystectomy treacherous. An aberrant right sectoral duct [often the right posterior sectorial duct] can sometimes empty directly into the gallbladder or cystic duct instead of the common hepatic duct. Such a duct can be easily misidentified as the cystic duct and mistakenly divided [19].
An important anomaly is the presence of accessory or aberrant bile ducts. Small subvesical bile ducts [often called ducts of Luschka] can connect liver segments directly to the gallbladder bed; minor ones are present in up to 12–50% of people [20]. While they usually are tiny (<1– 2 mm) and not seen intraoperatively, if one is relatively large and gets clipped or damaged, it can cause a postoperative bile leak. About 0.2–2% of cholecystectomies incur bile leaks, often from a cystic duct stump or an unrecognised duct of Luschka [21, 22]. Indeed, the most common mechanism of major bile duct injury is misidentification-e.g. confusing the common bile duct or an accessory duct for the cystic duct in an inflamed field. Low insertion of the cystic duct or a parallel course of the cystic duct can also cause inadvertent cut into the common duct. Routine use of landmarks like the Rouviere’s sulcus and techniques like intraoperative cholangiography or intraoperative usages of Indocyanine Green (ICG) can help in better visualisation of the ductal anatomy thus allowing identification of anomalies.
Mirizzi’s syndrome is a rare complication of gallstone disease where a stone becomes impacted in the cystic duct or Hartmann’s pouch of the gallbladder and compresses the common hepatic duct. Chronic compression can lead to erosion and formation of a cholecysto-biliary fistula between the gallbladder and common bile duct. Mirizzi syndrome is uncommon (occurring less than 1% in Western countries) [23]. Its presence dramatically increases the risk of bile duct injury if unrecognised and has a reported bile duct injury rates up to 17% when Mirizzi is undiagnosed at surgery [24]. Preoperative diagnosis can be challenging because the presentation can mimic ordinary choledocholithiasis or cholecystitis, thus identified in only 8–62% of cases [25]. Therefore, it is crucial to have a high index of suspicion in patients with long-standing gallstones who present with obstructive jaundice, cholangitis or a shrunken gallbladder with an impacted stone in the Hartmann’s pouch on imaging.
The Csendes classification is the most widely used system to categorize Mirizzi syndrome and is based on the presence and extent of cholecysto-biliary fistula and any cholecysto- enteric fistula [Figure 1] [26].
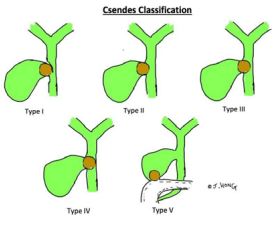
Figure 1.Diagrammatic representation of Csendes Classification
Types I and II are the most common presentations, whereas the higher types are relatively rare [23]. Mirizzi’s syndrome is usually diagnosed on imaging such as MRCP, CT scan or ultrasound. Mirizzi’s syndrome should be suspected if the patient has obstructive jaundiceplus gallstones without a clear common duct stone, or if imaging shows a stone in an infundibular position with bile duct dilation upstream. Intraoperatively, finding a gallstone eroding into the duct or an unusually large inflammatory mass at Calot’s triangle signals a Mirizzi’s.
Management depends on the type: For Type I [external compression without fistula], a careful subtotal cholecystectomy or even cholecystectomy can often be done, removing the gallstone and gallbladder while avoiding injury to the bile duct, sometimes combined with a cholangiogram to confirm duct patency. For Types II–IV [fistulas], the operation can involve a bile duct repair. Small fistulas may be managed with choledochoplasty using a flap of cystic duct or gallbladder remnant to close the defect. Some has proposed a self-expanding metal stent via ERCP for small bile duct defects [27, 28]. Larger defects might require biliary reconstruction such as a hepaticojejunostomy. Because the tissue is inflamed and scarred, these are high-risk cases for bile duct injury or stricture. Some authors recommend open surgery for higher-grade Mirizzi’s syndrome, or at least a very low threshold to convert from laparoscopy to open, due to the advanced dissection and suturing often required. If a cholecystoenteric fistula (Type V) is present, one must also address the intestinal fistula and be vigilant for gallstone ileus in the bowel.
Knowing the classifications helps in anticipating the required surgical steps and risks. Crucially, if Mirizzi syndrome is encountered or suspected, subtotal cholecystectomy could be performed, identification of the bile duct and fistula extent, and often performing an intraoperative cholangiogram or using choledochoscopy to evaluate the common duct. A difficult Mirizzi’s case should prompt consideration of involving hepatobiliary services for ongoing management.
Safe technique and a systematic approach are crucial when performing a cholecystectomy in difficult conditions. The primary goal is to avoid bile duct or vascular injuries while successfully removing the gallbladder. A “culture of safety” in cholecystectomy has been promoted by surgical societies [e.g. SAGES Safe Cholecystectomy program] to systematize the practice of safe techniques.
In summary, the best practices for difficult cholecystectomy center around strict adherence to safe dissection principles, excellent visualization, liberal use of intraoperative imaging, and the courage to halt and choose a safer alternative when anatomy is uncertain.
Bailout strategies are considered when conventional dissection cannot be safely performed to avoid catastrophic injury. These techniques aim to resolve the acute problem [remove the septic gallbladder or decompress it] while avoiding blind dissection in dangerous areas. The main bailout options include subtotal cholecystectomy, the fundus-first [dome-down] technique, and conversion to open surgery. In extreme cases, aborting the cholecystectomy and performing a cholecystostomy [drainage of the gallbladder] may be the safest approach. Each strategy is chosen based on intraoperative findings and the operator’s judgment.
Subtotal Cholecystectomy (STC) involves intentionally not removing the entire gallbladder. Indications for STC are a hostile Calot’s triangle where the cystic duct and artery cannot be safely isolated due to inflammation or fibrosis. The gallbladder can be truncated at the body/fundus and leaving the portion of gallbladder neck/cystic duct in situ which will markedly reduce the risk of tearing the common bile duct or hepatic artery. Two main variants are described:
Both approaches avoid dissecting in the hazardous area of the porta hepatis and a drain should be left in-situ before bailing out.
Subtotal cholecystectomy has become one of the most important bailouts in difficult cholecystectomy. Evidence shows that STC dramatically lowers the risk of bile duct injury compared to persisting with a total cholecystectomy in severe inflammation [34]. It can almost always be performed laparoscopically, thus obviating the need for a large open incision in many cases. Recent literature suggests that subtotal cholecystectomy is the single most effective bailout procedure currently for difficult gallbladders [33]. A 2016 HPB analysis succinctly concluded that the morbidity from bile duct injury far exceeds the morbidity from a subtotal cholecystectomy, firmly establishing STC as a reasonable and safe bailout strategy [35]. Bile leakage from the stump is a known complication, especially with the fenestrating technique, so a drain is typically left and the cystic duct may be secured with a suture or endoloop if possible. Patients should be informed that a remnant gallbladder remains, which in rare cases can cause a remnant cholecystitis. However, long- term follow-up of subtotal cholecystectomy patients is generally positive: the vast majority [over90%] have symptom resolution and avoid re-operation [36].
The fundus-first technique [also called dome-down cholecystectomy] is both a strategy for difficult cases and a potential step in a bailout. The gallbladder is dissected from the liver bed at the fundus and dissected down towards the cystic duct. This allows early identification of the cystic duct and artery when extensive scarring is present at Calot’s triangle. The fundus-first dissection could allow gallbladder to be dissected except for a small stump, intentionally converting the situation into a subtotal cholecystectomy. Key step is to stay in the correct tissue plane during dissection – the plane between the gallbladder serosa and the cystic plate of the liver. Large vessels of the right portal pedicle or a hole into the common duct from the side could be created if dissection goes into liver parenchyma or too deep toward the hilum.
In many difficult cholecystectomies, fundus-first approach is often used in tandem with subtotal cholecystectomy. Thus, fundus-first is an important tool in the arsenal and can be considered a form of “bailout” itself – an alternative dissection route that avoids the most inflamed area initially [37]. In summary, the dome-down technique is a valuable strategy in difficult gallbladders, often leading to a controlled subtotal resection depending on the intra-operative assessment.
Conversion to an open cholecystectomy remains an ever-present bailout option and is indeed the original fail-safe plan in laparoscopic surgery. Every Laparoscopic Cholecystectomy carries a risk of conversion [a few percent in elective cases, but significantly higher in acute or difficult cases]. Hesitation should not be made where laparoscopy does not allow safe progress due to severe inflammation, unclear anatomy, excessive bleeding, or other technical impasses.
Studies have shown that conversion rates in difficult cholecystectomy vary widely depending on the threshold of the surgeon and the patient population. Experienced minimally invasive surgeons, using bailouts like subtotal resection, have reported very low conversion rates [38]. The introduction of bailout techniques like laparoscopic subtotal cholecystectomy has reduced the need for open conversion in many centers, since the surgeon can avoid the dangerous dissection without needing to abandon laparoscopy [39]. Conversion to open is only beneficial if the procedure can be completed safely. Therefore, it is recommended that every surgeon should be adept at open biliary surgery or have a colleague available who is. If a surgeon is not comfortable with complex open biliary dissection [for example, a laparoscopy-trained surgeon who rarely does open cases], involvement of an HPB (hepatopancreatobiliary) specialist is wise either intraoperatively or by early referral.
Modern technology and evolving techniques are continually improving the safety of cholecystectomy, especially in difficult scenarios. Here we discuss a few recent advances and adjuncts that are particularly relevant: robotic-assisted cholecystectomy, routine or selective intraoperative cholangiography, Indocyanine Green (ICG) fluorescence guidance and artificial intelligence.
1. Robotic-Assisted Cholecystectomy:
Robotic surgical systems have been increasingly applied to cholecystectomy in the recent years. The robotic platform offers high-definition 3D visualisation and wristed instruments that enhance dexterity compared to straight laparoscopy, which could be advantageous in dense adhesions or awkward anatomies. Some reports suggest that robotic cholecystectomy can overcome difficulties related to visualisation and instrument maneuverability in difficult gallbladders, potentially reducing conversion rates in acute cholecystitis cases. For example, a recent study in an emergency setting found robotic cholecystectomy had a significantly lower risk of conversion to open surgery compared to laparoscopic in similar patients [40]. However, the data on improved outcomes with robotics are mixed. Smaller series have found the robotic approach safe and feasible in complex cases, but a large analysis of Medicare data reported a higher incidence of bile duct injury with robotic cholecystectomy as compared to standard Laparoscopic Cholecystectomy [41, 42]. This might reflect the learning curve and early adoption phase; as robotics becomes more widespread, outcomes may improve. At present, robotic cholecystectomy is generally considered as effective as laparoscopy for gallbladder disease, with the potential for improved surgeon ergonomics and precision in difficult cases. The downsides include higher cost, longer setup time, and limited availability in many centers. The consensus is that robotics is a promising adjunct for difficult cholecystectomy but not an absolute necessity.
The decision to use robotics often comes down to surgeon preference and resource availability. robotic cholecystectomy is an emerging tool that can be considered in difficult cases, showing some trend toward lower conversion, but its impact on bile duct injury rates is not clearly proven and vigilance is still required.
2. Intraoperative Cholangiography [IOC]:
Intraoperative cholangiography is a classic adjunct rather than a new one, but it continues to be a topic of debate and improvement in the context of difficult cholecystectomy. IOC involves cannulating the cystic duct and injecting contrast to visualize the biliary tree under fluoroscopy during surgery. Its utility in difficult cases allow anatomic clarification and detection of bile duct stones or injury-IOC can identify unsuspected common bile duct stones [in roughly 4% of cases, stones are found on IOC in some series], allowing them to be dealt with in the same setting (via CBD exploration or postoperative ERCP) [43]. It can also show a leak or extravasation if a partial injury has occurred, enabling immediate repair.
Guidelines by organizations like SAGES and WSES support liberal use of IOC, particularly in high-risk cases. A large meta-analysis by Donnellan et al. of 62 studies concluded IOC is a useful tool with a high detection rate of abnormalities and can be done selectively based on risk factors [43]. Critics of routine IOC note it can prolong operative time and that there is no Level-I evidence proving it reduces the incidence of bile duct injury. However, some population studies have observed lower BDI rates at institutions with a policy of routine IOC, suggesting it may serve as a safeguard. Cholangiography remains a widely used adjunct to enhance safety, and every difficult cholecystectomy should include consideration of an IOC at the very least. In some healthcare systems, IOC is done routinely on all cholecystectomies as standard practice.
3. ICG Fluorescence Cholangiography:
A notable recent advance in biliary surgery is the use of indocyanine green [ICG] fluorescence imaging to visualize bile ducts intraoperatively. ICG is a nontoxic dye that, when injected intravenously [usually 0.25– 0.5 mg/kg a few hours or minutes before surgery], is taken up by the liver and excreted into bile. Using a near-infrared [NIR] camera system on laparoscopic or robotic equipment, the biliary tree can be seen glowing [fluorescing] in real-time during the operation. Several studies suggest that ICG enhances identification of extrahepatic bile ducts and can help avoid misidentification injuries [44-46]. Notably, unlike an IOC which is typically done after dissection has started, ICG can outline ducts before any dissection – sometimes the cystic duct is seen entering the common duct as a fluorescent structure. A systematic review of ICG use found that it reduced conversion rates (0.5% vs 2.5% in non-ICG cases) and had a lower incidence of bile duct injury (0.12% vs 1.3%) in a pooled analysis [45]. Another study in patients with acute cholecystitis after percutaneous gallbladder drainage showed ICG fluorescence guidance significantly shortened operative time and drastically lowered conversion to open (2.6%vs 22% without ICG) [47]. These are promising results, indicating that fluorescence cholangiography can be a real asset in difficult cases by making the invisible anatomy visible. Early studies and meta- analyses have shown reductions in operative time, conversion, and possibly bile duct injury when ICG is used.
4. Artificial Intelligence:
Although in early stages, research is underway on machine learning tools that could, for example, highlight biliary anatomy on the surgeon’s screen automatically or warn when dissection is nearing a critical structure. Some experimental systems already attempt automatic identification of the cystic duct/artery on the laparoscopic feed [48, 49]. While not yet in clinical use widely, these technologies represent the next frontier in preventing errors.
Challenging cholecystectomy scenarios demand a high level of surgical awareness, flexibility, and adherence to safe principles. Surgeons must know their hepatobiliary anatomy, employ sound intraoperative strategies, and be willing to adjust the operative plan when managing severe cholecystitis, aberrant anatomy, or other challenging gallbladder conditions. Key steps include obtaining the critical view of safety, utilizing cholangiography or modern fluorescence techniques to clarify anatomy, and recognizing when to bailout. With continued emphasis on education, research, and adoption of best practices worldwide, the management of challenging gallbladder cases will continue to improve, hopefully driving complications and conversion rates ever lower.